- English
- French
- German
- Portuguese
- Spanish
- Russian
- Japanese
- Korean
- Arabic
- Greek
- German
- Turkish
- Italian
- Danish
- Romanian
- Indonesian
- Czech
- Afrikaans
- Swedish
- Polish
- Basque
- Catalan
- Esperanto
- Hindi
- Lao
- Albanian
- Amharic
- Armenian
- Azerbaijani
- Belarusian
- Bengali
- Bosnian
- Bulgarian
- Cebuano
- Chichewa
- Corsican
- Croatian
- Dutch
- Estonian
- Filipino
- Finnish
- Frisian
- Galician
- Georgian
- Gujarati
- Haitian
- Hausa
- Hawaiian
- Hebrew
- Hmong
- Hungarian
- Icelandic
- Igbo
- Javanese
- Kannada
- Kazakh
- Khmer
- Kurdish
- Kyrgyz
- Latin
- Latvian
- Lithuanian
- Luxembou..
- Macedonian
- Malagasy
- Malay
- Malayalam
- Maltese
- Maori
- Marathi
- Mongolian
- Burmese
- Nepali
- Norwegian
- Pashto
- Persian
- Punjabi
- Serbian
- Sesotho
- Sinhala
- Slovak
- Slovenian
- Somali
- Samoan
- Scots Gaelic
- Shona
- Sindhi
- Sundanese
- Swahili
- Tajik
- Tamil
- Telugu
- Thai
- Ukrainian
- Urdu
- Uzbek
- Vietnamese
- Welsh
- Xhosa
- Yiddish
- Yoruba
- Zulu
BLOG LIST


To get around in the bulk L-Carnitine market, you need to know about the quality standards, supplier qualifications, and formulation needs that make supplement goods work. L-Carnitine Pure Powder is an important nutrient for weight loss and energy metabolism products, so it's important to choose a seller carefully and check the quality. This complete guide solves all the problems that supplement brands have with buying things. It talks about quality standards, following the rules, and finding cheap sources to make your product development and production processes run more smoothly.
Understanding Bulk L-Carnitine Pure Powder
As a naturally occurring amino acid product, L-carnitine has the molecular formula C₇H₁₅NO₃ and a molecular weight of 161.2. It is a white solid powder that is 98% pure. This chemical is very important for making energy in cells because it moves fatty acids to mitochondria to be burned, turning them into useful energy while also helping the body burn fat and keep its energy balance.
Chemical Properties and Specifications
The Chemical Abstracts Service gives L-carnitine the CAS number 541-15-1, which is a common way to identify it for quality control and regulatory reasons. High-quality bulk powder has a uniform solid structure and meets strict purity standards that can be checked using HPLC. Products are usually packed in 1 kg metal foil bags or 25 kg drums by factories to keep them stable and clean while they are being stored and shipped.
Metabolic Functions and Applications
Long-chain fatty acids can move more easily across mitochondrial membranes with the help of L-carnitine. This makes energy generation more efficient at the cellular level. When mixed with the right diet and exercise plans, this process helps people who are trying to lose weight by stopping body fat from building up. These metabolic benefits are used by supplement companies to make specific formulas for fitness fans, weight loss programs, and general health uses.
Product Variants and Formulation Considerations
Depending on the purpose and audience, different types of L-carnitine offer different benefits. Acetyl L-carnitine is better absorbed by the body and can cross the blood-brain barrier more easily, which makes it a good choice for cognitive support products. L-carnitine tartrate is more stable as a powder and dissolves better, while fumarate versions add extra biochemical cofactors that might make the product work better overall in some situations.

Criteria for Selecting Bulk L-Carnitine Pure Powder Suppliers
For supplier relationships to work, quality standards, legal compliance, and managerial skills that make sure products like L-Carnitine Pure Powder are delivered consistently must all be carefully looked at. Reliable providers keep a number of certificates, such as FDA registration, organic certification, kosher certification, ISO certification, HALAL certification, HACCP certification, and GMP standards. These show that they are committed to quality and following the rules.
Essential Certifications and Quality Standards
Manufacturing plants should follow GMP guidelines and have separate labs for checking the quality of their products and specialized production lines. Usually, these places have controlled settings with the right ways to keep things clean and keep an eye on stability from batch to batch. Certified suppliers of organic, kosher, and halal goods open up new market possibilities and meet the needs of a wide range of customer groups.
Testing Protocols and Purity Verification
HPLC testing methods accurately measure the amounts of active compounds and find any possible contaminants or flaws that might lower the quality of the product. Suppliers you can trust test for heavy metals, microbial pollution, and leftover solvents, and they keep thorough certificates of analysis for each production batch. These testing procedures make sure that international standards are followed and help with regulatory applications in many markets.
Pricing Structures and Payment Options
Competitive sellers offer a range of payment options, such as Western Union, Alipay, XT, and telegraphic transfer, so that they can handle foreign sales and cash flow needs. Bulk prices usually get better as you order more, and the 1 kg minimum order size lets smaller brands get high-quality products without having to keep too much inventory on hand. Delivery times of 3–7 working days after payment confirmation make it possible to plan production and keep track of supplies more efficiently.
Comparing Bulk L-Carnitine Powder with Alternative Forms and Brands
Choosing the right product format has a big effect on how quickly and cheaply supplements can be made, as well as how stable the recipe is and how well it works. Bulk powder forms are more flexible than pre-encapsulated ones because they let you precisely control dosing and create custom formulations that fit your brand's positioning and the needs of your customers.
Powder Versus Capsule and Liquid Forms
Bulk powder gives supplement makers the most formulation options, so they can make their own mixes, change the strength levels, and add ingredients that work well together for enhanced benefits. When kept properly in a dry, cool place, this format usually saves you money compared to pre-made pills and keeps your medicine fresh longer. Powder forms also make private label production easier and let brands come up with unique ways to show their products that set them apart from competitors in the market.
Storage and Stability Considerations
When stored properly in covered cases in dry, cool places, the shelf life can be extended to two years while the product stays potent and doesn't break down. Aluminum foil packing keeps food from absorbing moisture and rusting, which could lower the quality of the food over time. Controlling the temperature during storage and shipping is very important to keep the crystal structure and stop clumping or discoloration that can change how the product looks or how well it works.
Cost-Benefit Analysis for Different Suppliers
Established suppliers with a history of trustworthiness usually charge more, but they offer benefits like consistent quality, help from regulators, and a reliable supply chain. While newer suppliers may offer competitive prices, you will need to do more research on their quality systems and output skills. The total cost of ownership includes not only the price of the ingredients but also the costs of quality control, help with legal compliance, and the risk of a product recall, which could have a big effect on the brand's image and its ability to make money.

Best Practices for Using Bulk L-Carnitine Pure Powder in Supplement Manufacturing
Formulation strategies that work best improve the bioavailability and customer happiness while also making sure that all market groups are safe and following the rules. Clinical study supports daily doses of L-Carnitine Pure Powder ranging from 500 mg to 3000 mg, based on the intended use and target population. Formulations for weight loss usually use higher concentrations than general health goods.
Formulation Guidelines and Dosage Recommendations
Weight loss pills often have 1000 to 2000 mg in each dose to help the body's metabolism and energy production while exercising or limiting calories. Sports nutrition items may use similar doses and suggest times to take them that match how people normally eat before or after working out. In general health formulas, lower amounts of 500 to 750 mg are used to support normal metabolic function without overstimulating it or giving the user too much energy.
Quality Control and Manufacturing Standards
To make sure that all of the ingredients are evenly distributed in the finished product, the manufacturing process should include detailed mixing steps. This is especially important when mixing with other active ingredients or excipients. Controlling the moisture during processing stops the product from breaking down and keeps it stable for as long as it's on the shelf. Testing final goods on a regular basis makes sure that promises about their potency are true and finds any possible reactions with other ingredients in the mixture that might change their bioavailability or stability.
Regulatory Compliance and Labeling Requirements
Labeling that is done right includes correct lists of ingredients, suggestions for portion sizes, and following FDA rules for making food supplements. When making the first formulation, you should think about whether international markets need any extra licenses or testing procedures. As part of the documentation needs, there must be thorough records of the manufacturing process, certificates from suppliers, and test results for each batch that can be used for regulatory reports and quality assurance programs.
How to Procure Bulk L-Carnitine Pure Powder Efficiently?
Strategic buying methods get the best value for money while also making sure quality is always the same, and the supply chain works reliably, including for L-Carnitine Pure Powder. Building relationships with certified sellers who keep enough goods on hand and offer flexible shipping times keeps production from stopping and helps businesses reach their growth goals in a variety of market situations.
Supplier Evaluation and Selection Process
A full evaluation of a seller includes checking out the building, the quality system, and references from current customers in similar markets. Suppliers should show that they have enough production capacity, backup methods for important tools, and plans for what to do if there are problems with the supply chain. A review of financial stability makes sure that the relationship will last for a long time and lowers the risks that come with a provider going bankrupt or making changes to their operations that could affect the availability of their products.
Sample Testing and Quality Verification
The first review of a sample tells us a lot about the quality, consistency, and ability of the product to work with current formulation methods. Identity verification, purity analysis, and stability studies should all be part of the testing process. These should be done in a variety of storing conditions that are similar to those found in real life. Multiple samples from different batches help find problems with possible variation and set quality standards that are used to keep an eye on the supplier's performance.
Long-Term Partnership Development
Setting up relationships with chosen suppliers lets you get bulk discounts, priority handling during shortages, and chances to work together on product development that helps your brand stand out. Talking about market trends, changes to regulations, and new technologies on a regular basis helps businesses stay ahead of the competition and spot new opportunities. When you're negotiating a contract, you should talk about quality standards, delivery dates, and performance measures that are in line with your business goals and the needs of risk management.
Conclusion
To successfully buy L-carnitine, you need to find a balance between high standards, following the rules, and cost factors that support long-term business growth. Brands of supplements can benefit from working with certified providers who can consistently deliver high-quality goods and provide detailed paperwork and technical know-how that helps with product development. Some of the most important factors are carefully evaluating suppliers, following the right steps for quality control, and managing relationships in a way that guarantees steady access to high-purity ingredients for developing competitive formulations.
FAQ
1. What distinguishes high-quality L-carnitine pure powder from inferior alternatives?
The 98% purity of premium L-carnitine pure powder is confirmed by HPLC testing methods, and the powder always has a white, crystalline look and the right chemical structure. Good goods come from factories that are GMP-certified and have a lot of paperwork, like certificates of analysis, heavy metal tests, and proof that the products are free of microbes.
2. How can supplement brands verify product quality before bulk purchases?
Ask for specific certificates of analysis, have samples tested by a third party, and check the certifications of suppliers through government databases. Identity testing, purity analysis, and stability tests should all be part of quality verification. These tests should be done in a variety of storage conditions that are similar to those found in production and distribution settings.
3. What regulatory considerations apply when importing L-carnitine for supplement manufacturing?
The main legal issues that need to be thought about are FDA registration standards, import paperwork, and following current good manufacturing practices. International suppliers should keep the right licenses, such as an FDA facility registration, and goods must follow the labeling rules for dietary supplements and the safety rules set by the proper regulatory authorities.
4. What minimum order quantities and delivery timeframes should brands expect?
Suppliers you can trust usually have minimum orders that start at 1 kg and arrival times that range from 3 to 7 working days after payment is confirmed. Larger orders may be eligible for bulk savings and faster shipping choices. On the other hand, smaller orders allow for initial testing and recipe development without having to commit to too much inventory.
5. How do different L-carnitine forms compare for supplement applications?
L-carnitine tartrate is very stable and easy to dissolve, making it a good choice for powder forms. Acetyl L-carnitine, on the other hand, is better for brain support products because it is more bioavailable. Pure L-carnitine base gives formulators the most options and the best value for money when making weight loss and sports nutrition products.
Contact Jianbei for Premium L-Carnitine Pure Powder Supply Solutions
Pharmaceutical-grade L-carnitine pure powder is available from Jianbei Biotechnology to support your supplement brand. This powder is backed by full quality assurance and legal compliance. Our manufacturing center is GMP-certified and has three specialized production lines, independent labs, and a large inventory to make sure that products are always available. As a reputable company that makes L-Carnitine Pure Powder, we offer full certifications that meet foreign standards, such as FDA, ISO, HALAL, and organic certifications. Email our purchasing agents at sales@bqingbio.com to get samples, get low prices, and talk about custom recipe solutions that can speed up the development of your product while still meeting the highest quality standards.
References
1. Flanagan, J.L., et al. "Role of carnitine in disease prevention and treatment." Nutrition in Clinical Practice, 2010, 25(2), 148–159.
2. Evans, A.M., and Reuter, S.E. There was an article in Clinical Pharmacokinetics (2012), 51(9), 553-572, called "Carnitine and acylcarnitines: pharmacokinetic, pharmacological and clinical aspects."
3. Hongu, N., and Sachan, D.S. "Adding carnitine and choline supplements along with exercise changes the levels of biochemical markers of fat metabolism and serum leptin in healthy women." Journal of Nutrition, 2003, 133(1), 84–89.
4. Karlic, H., and Lohninger, A. "Supplementation of L-carnitine in athletes: does it make sense?" Nutrition, 2004, 20(7-8), 709–715.
5. Ruggenenti, P., et al. "Ameliorating hypertension and insulin resistance in subjects at increased cardiovascular risk: effects of acetyl-L-carnitine therapy." Hypertension, 2009, 54(3), 567–574.
6. Stephens, F.B., et al. "New insights concerning the role of carnitine in the regulation of fuel metabolism in skeletal muscle." Journal of Physiology, 2007, 581(2), 431–444.

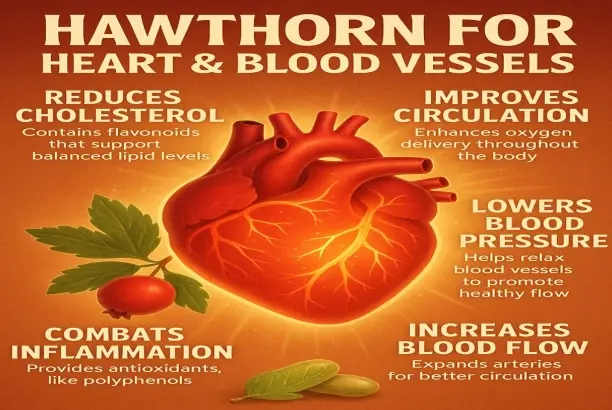
Hawthorn powder serves as a versatile botanical ingredient derived from Crataegus species, offering manufacturers exceptional opportunities to develop cardiovascular health supplements and functional foods. This natural extract contains bioactive compounds, including flavonoids, oligomeric proanthocyanidins, and organic acids that support heart health, digestive function, and antioxidant activity. The powder form provides excellent stability and ease of incorporation into various product formats, making it ideal for capsules, tablets, functional beverages, and nutritional bars. With its light pink appearance and standardized 98% purity specifications, hawthorn powder enables formulators to create consistent, high-quality products that meet regulatory requirements across global markets.
Understanding Hawthorn Powder and Its Functional Benefits
One of the plant ingredients that has been studied the most for its effects on heart health is hawthorn juice. Traditional medicine has used plants in the Crataegus family for hundreds of years. Modern research has shown that these plants can be used as medicine, with clinical tests showing measurable health effects.
Cardiovascular Support Mechanisms
The main beneficial chemicals in hawthorn extract are related to vitexin, hyperoside, and quercetin. There are several ways that these flavonoids work together to help the heart operate. According to research, these chemicals help keep blood pressure at a healthy level by widening blood vessels and boosting healthy blood flow. The molecular formula C16H14O4 with a molecular weight of 270.28 is the normal active ingredient that always has the same medicinal effects.
Regularly taking hawthorn extract may help lower amounts of low-density lipoprotein (LDL) cholesterol and improve heart health in general, according to clinical research. These flavonoids' antioxidant qualities protect heart cells from oxidative stress, which is a key part of keeping the heart healthy over time.
Digestive Health Benefits
In addition to helping the heart, hawthorn powder has organic acids and enzymes that make stomach juices flow more easily, which makes digestion better. This system helps ease bloating and loss of hunger while making the digestive system more comfortable overall. Because it is good for digestion, hawthorn powder is very useful for functional food uses that aim to improve gut health.
Quality Specifications and Testing
Premium hawthorn extract goes through strict HPLC testing to make sure it is 98% pure. This analytical method gives exact amounts of active chemicals, which is necessary for making supplements because it ensures stability from batch to batch. The Chemical Abstracts Service ID 36052-37-6 makes it easy to identify legal documents and quality control processes.
Selecting the Right Hawthorn Powder for Your Supplement or Functional Food Product
To pick the best hawthorn extract, you need to carefully consider a number of factors that affect how well the product works, how well it meets regulations, and how efficiently it is made, including hawthorn powder. To make the best decisions, you have to find the best balance between quality requirements, provider skills, and cost factors.
Quality Standards and Certifications
Some of the certifications that high-quality hawthorn extract should have are FDA registration, organic certification, kosher certification, ISO compliance, HALAL compliance, HACCP compliance, and GMP compliance. These certificates make sure that the ingredient meets the high standards of quality needed for foreign sales. Quality control is important for pharmaceutical-grade uses, and GMP-certified factories with separate labs and specialized production lines can provide it.
You can't say enough good things about thorough testing. Quality testing should include more than just HPLC analysis for active chemicals. It should also include heavy metal screening, bacterial testing, and analysis of pesticide residues. These factors make sure that products in all market groups are safe and follow the rules.
Physical Characteristics and Formulation Compatibility
High-quality hawthorn extract looks like a light pink powder, which means it was processed and standardized correctly. This constant coloration shows the best conditions for extraction, which keep the bioactive chemicals while keeping the stability. You can easily flow and mix the powder form, which is very important for large-scale industrial processes.
The amount of moisture and the spread of particle sizes have a big effect on how well a mixture works. When hawthorn extract is handled correctly, it keeps its low moisture levels, which keeps it from clumping and helps it last longer. Quality hawthorn extract is naturally stable, as shown by the fact that it can be kept for two years in a dry, cool place.
Supplier Evaluation Criteria
Partnering with a source that works well depends on more than just price. Long-term success is closely linked to production capacity, quality processes, and technical support skills. Suppliers who run GMP-compliant workshops with various production lines show that they have the means to keep up with rising demand.
With a minimum order quantity of 1 kilogram, product creation can be done with more freedom, and operations can run smoothly. The shipping period of 3–7 working days makes it possible for flexible supply chain management, which is necessary to keep production schedules.
Best Practices for Incorporating Hawthorn Powder into Supplements and Functional Foods
In order to effectively add hawthorn extract to finished goods, you need to know about formulation principles, stability issues, and processing needs. These things affect how well a product works, how long it lasts, and how well customers like it.
Dosage Optimization and Bioavailability
Based on the application and health benefits desired, clinical studies show that the best daily dose is between 300 mg and 900 mg. Higher doses are usually needed for cardiovascular support, while smaller doses may work for digestive health uses. The 98% pure requirement lets you accurately figure out dosages and mark products in a consistent way.
One way to improve bioavailability is to mix hawthorn powder with other ingredients that help the body absorb it. Some vitamins and minerals can help the body absorb flavonoid molecules better, which makes the product work better overall. Formulators can make better goods with more benefits for customers if they understand these synergistic connections.
Formulation Techniques and Processing Considerations
The security of hawthorn extract is very good under normal processing settings. The powder form works perfectly with many production processes, such as straight compression tableting, capsule filling, and beverage use. To keep bioactive chemicals safe during heat-intensive processes, temperature sensitivity needs to be taken into account.
Encapsulation methods keep delicate chemicals from breaking down in the environment and allow for controlled release patterns. In functional food uses, new encapsulation ways can make the food more stable and taste better. In most products, hawthorn extract's natural taste profile doesn't need much masking.
Packaging and Storage Requirements
When hawthorn extract is properly packed, it stays safe from moisture, light, and air that can break down the active chemicals. For smaller amounts, aluminum foil bags work great as a shield. For big storage, fiber drums are a cost-effective way to protect things. UV-protective packaging keeps light-sensitive flavonoids from breaking down.
The stability and shelf life of a product are greatly affected by how it is stored. Keeping the product in a dry, cool place below 25°C with a relative humidity of less than 60% will keep its quality for two years. These keeping rules are in line with how supplements and food ingredients are usually handled.

Procurement Strategies in Bulk: Ensuring Cost-Effective Supply Without Compromising Quality
When buying hawthorn extract or hawthorn powder strategically, you have to balance a lot of factors, such as cost, quality, supply security, and following the rules. Procurement methods that work well take these things into account and support long-term business goals.
Supplier Relationship Management
Building good relationships with suppliers gives you a competitive edge that goes beyond price. When you work directly with makers, you can better control quality, get expert help, and see what's going on in the supply chain. Audits and quality reviews of facilities on a regular basis make sure that they are always in line with changing government rules.
Long-term arrangements allow for savings based on volume and protect supply during market changes. Specifications for quality, release dates, and pricing systems that protect both sides' interests should be included in these contracts. International business needs payment options that are easy to use, like telegraphic transfer, Alipay, PayPal, and Western Union.
Market Analysis and Pricing Strategy
The market for hawthorn extract has steady prices that change with the seasons based on the supply of raw materials. By understanding how these markets work, you can make smart buying decisions that keep supplies going while minimizing costs. Materials that are approved organic or pharmaceutical grade cost more because they have to go through more testing and processing.
When you buy in bulk, you get bigger discounts on prices, and the biggest price cuts usually happen when you buy 100 kilograms or more. But storage costs and managing goods must be taken into account when weighing possible savings against the need for operating capital.
Risk Management and Supply Chain Security
Supplier networks with a lot of different suppliers protect against delivery problems and keep prices low. Having a lot of qualified sources lets you make sourcing plans that are flexible and can adapt to changing market conditions. Suppliers are checked on a regular basis to make sure they are still able to meet quality standards.
Geographic diversity lowers the risks that come with area problems like bad weather, new rules, and transportation problems. Keeping smart amounts of goods on hand gives you a buffer stock while lowering the risks of quality degradation and carrying costs.
Case Studies: Successful Integration of Hawthorn Powder in Functional Food and Supplement Products
Real-life uses show that hawthorn extract has business promise in a wide range of product categories. These examples show formulation methods, market positioning, and factors that affect customer acceptance that work well.
Cardiovascular Health Supplements
A well-known supplement company made a complete heart health product with hawthorn juice as the main active ingredient. The mixture included 600 mg of pure hawthorn extract along with CoQ10, magnesium, and B vitamins to work together. Clinical studies that backed up the ingredient claims helped the company get strong marketing roles and suggestions from doctors.
Targeted marketing to health-conscious customers and healthcare professionals helped the product get a big share of the market. Premium pricing tactics were backed by quality standards like organic and non-GMO verification. According to customer comments, the main benefits were more energy and better circulatory health in general.
Functional Beverage Applications
A creative beverage business added hawthorn extract to a functional tea that is ready to drink and is meant to help with gut health. The recipe combined the naturally sour taste of hawthorn with flavors and herbs that went well with it. Bioactive chemicals were kept while industrial shelf stability was achieved using advanced processing methods.
A market study showed that consumers strongly agreed with the digestive health stance. People in the target audience who were looking for clean-label alternatives to manmade chemicals related to the story of the natural ingredients. Retail relationships with natural food channels were a good way to get into new markets and build brands.
Traditional Medicine Integration
Standardized hawthorn extract capsules were made for foreign markets by a pharmaceutical business that specializes in traditional Chinese medicine. The product passed strict quality standards for pharmaceuticals, such as USP guidelines and ICH stability guidelines. Regulatory approvals in a number of foreign areas were based on detailed documentation.
Strong evidence from clinical trials and traditional use history supported statements of effectiveness for heart and gut health with Hawthorn powder. The standardized extract format was appealing to doctors and nurses who wanted to suggest reliable, uniform goods to their patients.
Conclusion
Hawthorn powder is a great choice for vitamin and functional food companies that want to use ingredients that are backed by science and are popular with customers. A lot of studies have shown that this plant ingredient is good for your heart and digestive system. It also works well in formulations, which makes it a great choice for product development projects.
To be successful with hawthorn extract, you need to choose reliable providers who offer certified ingredients with full paperwork. When you combine historical use with modern scientific proof, you get great marketing chances for a wide range of consumer groups. Companies that put in the time to learn about manufacturing needs and how the market works will find that hawthorn extract is a good addition to their product lines.
FAQ
1. What is the recommended dosage range for hawthorn extract in supplements?
Depending on the health problem, clinical studies support daily doses of 300 mg to 900 mg of pure hawthorn extract. For cardiovascular support, 600 to 900 mg daily is usually enough. For gut health, 300 to 500 mg daily may be enough. For best absorption and effectiveness, these doses should be split into two to three daily meals.
2. How should hawthorn powder be stored to maintain quality?
Hawthorn extract needs to be kept in a cool, dry place that is below 25°C and has a relative humidity of less than 60%. Using the right packing in containers that keep out wetness saves the environment from damage. If you keep the extract properly, it will stay fully active for two years from the date it was made. Light-sensitive materials don't break down when they are protected from UV light.
3. What certifications should I look for when sourcing hawthorn extract?
A high-quality hawthorn extract should have several certificates, such as FDA registration, GMP compliance, ISO certification, and, if necessary, organic proof. Extra certificates like kosher, HALAL, and HACCP show that the quality management system is complete. These licenses help people get into foreign markets and make sure that rules are followed.
4. Can hawthorn extract be combined with other ingredients safely?
Including vitamins, minerals, and other plant products, hawthorn extract works very well with most supplement ingredients. Some common beneficial pairings are digestive enzymes for gut health and magnesium to support the heart. When making complicated goods with many ingredients, formulation testing should make sure the products are stable and bioavailable.
5. What quality testing methods verify hawthorn extract purity?
HPLC research is the best way to measure the amount of active chemicals in hawthorn extract, and it meets 98% purity standards. Heavy metal screening, microbial analysis, and chemical residue testing are some of the other tests that are done. For each batch, there should be a certificate of analysis to show that it meets the quality standards.
Partner with Jianbei for Premium Hawthorn Powder Solutions
Jianbei Biotechnology is a reliable company that makes hawthorn powder. They have advanced research and development (R&D) tools and GMP-certified production facilities that allow them to make high-quality plant extracts. Our large inventory and many certifications allow us to meet urgent shipping needs while keeping pharmaceutical-grade quality standards. We have the technical know-how and reliable supply chain needed for successful product creation. We have 15 national invention patents and the most up-to-date production facilities.
Our hawthorn extract is 98% pure, as proven by HPLC testing. It comes with full paperwork packages and a variety of payment methods, such as PayPal and telegraphic transfer. Get in touch with our team at sales@bqingbio.com to talk about custom formulations and mass prices. Get the Jianbei advantage by taking advantage of our dedication to cutting-edge technology and thorough quality control systems that are made to help your business grow.
References
1. Chang, Q., Zuo, Z., Harrison, F., & Chow, M. S. (2002). Hawthorn: a review of its clinical pharmacology and therapeutic uses in cardiovascular disease. Journal of Clinical Pharmacology, 42(6), 605-612.
2. Holubarsch, C. J., Colucci, W. S., & Eha, J. (2018). Benefit-risk assessment of Crataegus extract WS 1442: an evidence-based review. American Journal of Cardiovascular Drugs, 18(1), 25-36.
3. Zick, S. M., Gillespie, B., & Aaronson, K. D. (2008). The effect of Crataegus oxycantha special extract WS 1442 on clinical progression in patients with mild to moderate symptoms of heart failure. European Journal of Heart Failure, 10(6), 587-593.
4. Walker, A. F., Marakis, G., & Morris, A. P. (2002). Promising hypotensive effect of hawthorn extract: a randomized double-blind pilot study of mild, essential hypertension. Phytotherapy Research, 16(1), 48-54.
5. Daniele, C., Mazzanti, G., & Pittler, M. H. (2006). Adverse-event profile of Crataegus spp.: a systematic review. Drug Safety, 29(6), 523-535.
6. European Medicines Agency. (2016). Assessment report on Crataegus spp., folium cum flore and Crataegus spp., fructus. Committee on Herbal Medicinal Products, EMA/HMPC/159075/2014.

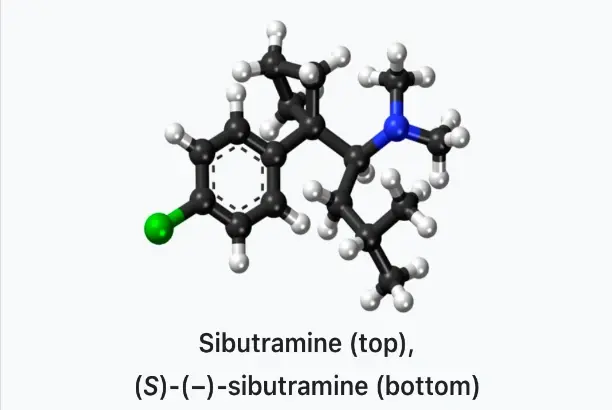
Sibutramine original powder is a strong hunger suppressant drug that is meant to help people lose weight by changing specific neurochemicals. This white, crystalline powder is 98% pure and works by changing chemical paths in the brain. It specifically targets serotonin and norepinephrine reuptake processes to improve signals of fullness and decrease signals of hunger. Aside from just making you feel less hungry, research shows that this substance may help improve metabolic signs like insulin sensitivity and lipid profile improvement when used with the right exercise and diet plans.
Understanding Sibutramine Original Powder: Benefits and Mechanism
The original powder form of sibutramine is a complex pharmaceutical substance with the formula C₁₇H₂₆ClN and a molecular weight of 279.85. The Chemical Abstracts Service gives this substance the CAS number 106650-56-0, which tells us exactly what it is chemically for quality control and legal reasons.
Pharmacological Action and Neurotransmitter Modulation
The main way it works is by blocking the release of serotonin, norepinephrine, and dopamine in brain synapses. This three-step method makes neurotransmitters available for a longer time, which leads to longer signs of fullness and less desire to eat. When this mechanism is part of complete weight management plans, clinical studies show that it helps people lose weight in a measured way.
The compound's solubility profile shows the best rates of absorption, which makes it very useful for formulation scientists who are making weight loss products. The factories that make this stuff usually have GMP-standard workplaces that are checked by an outside lab to make sure of the consistency of the material's strength and purity.
Metabolic Enhancement Properties
Aside from making you feel less hungry, sibutramine may also help your metabolism work better. Studies show that signs of insulin resistance improve and that changes in blood cholesterol patterns are good. These extra benefits make the substance appealing to companies that make supplements and are looking for weight control ingredients that can do more than one thing.
As part of quality control procedures for sibutramine original powder, HPLC testing is often used to confirm that it meets 98% pure standards. Compounds stay stable for up to two years when stored properly in dry, cool places and properly packed, which makes them good for bulk purchasing.
Comparison of Sibutramine Original Powder with Other Weight Loss Solutions
A study of the market shows that there are big differences between sibutramine original powder and other hunger suppressants that are currently on the market for makers and formulators. Understanding these distinctions enables informed procurement decisions for companies developing weight management products.
Purity and Bioavailability Advantages
Standard forms of sibutramine are 98% pure, which is higher than many generic options, which are usually only 85–92% pure. This higher level of purity means that dosing needs can be predicted more accurately, and formulations are more consistent for large-scale production.
In terms of absorption, high-purity sibutramine is better than natural hunger suppressants like garcinia cambogia or hoodia extracts. Some people like natural options, but the uniform potency of sibutramine gives formulators the exact dosing control they need to meet regulatory requirements and make claims about its effectiveness.
Cost-Effectiveness Analysis
If you buy original sibutramine powder in bulk, you can save money compared to other hunger suppressants. Because the minimum order amount is 1 kg, smaller producers can get this ingredient without having to keep a lot of stock on hand. Different people have different tastes when it comes to overseas purchasing. Payment options like telegraphic transfer, Alipay, PayPal, and Western Union make it easy for everyone to make purchases.
Another important benefit is that manufacturing can be scaled up or down. Companies that already have production lines can make sibutramine powder quickly and easily. On the other hand, plant options often need complicated extraction methods that raise costs and cause delays in production.
Procurement Insights: Buying Sibutramine Original Powder for B2B Clients
To successfully buy original sibutramine powder, you need to pay close attention to quality paperwork, supplier verification, and regulatory compliance procedures. International buyers have to deal with a lot of complicated certification rules while also making sure of the purity of the product throughout the supply chain.
Supplier Verification and Certification Requirements
Reliable providers keep a full set of certificates, such as FDA registration, ISO compliance, HALAL, HACCP, and GMP certifications. These certificates show that the company follows international quality standards, which are necessary for global marketing networks.
An evaluation of a manufacturing site shows important operational skills. The equipment needed for uniform quality output is provided by suppliers who run GMP-standard workshops with multiple production lines and separate labs. Three production lines provide flexibility and allow capacity to be increased or decreased to meet changing demand trends.
International Compliance and Documentation
Suppliers who focus on exporting know what paperwork is needed for deals that happen across borders. Using the right customs forms, certificate of analysis reports, and batch tracking paperwork makes the importing process go more smoothly in many legal areas.
International shipping keeps the quality of goods safe by using 1 kg metal foil bags or 25 kg drums as required by transportation packaging standards. These package requirements follow the rules for dangerous materials and keep the compound stable during long transport times.
Order Processing and Delivery Logistics
With streamlined order handling, goods are usually sent out 3–7 business days after payment is confirmed. This quick turnaround helps just-in-time inventory management techniques and keeps buying teams' working capital needs to a minimum.
Flexible minimum order amounts starting at 1 kg can be used for a wide range of business sizes, from small-scale R&D projects to full-scale commercial production of sibutramine original powder. Because the item is easy to get, smaller businesses can try it out before committing to buying more of it.

Practical Dosage Guidelines and Usage Recommendations
Manufacturers who want to use original sibutramine powder in business products need to follow industry-standard dosage frameworks. These suggestions strike a balance between the need for effectiveness and the need to follow the rules in a variety of market settings.
Commercial Formulation Considerations
Professional manufacturing practices stress the importance of gradually increasing dosages to help users build up a tolerance and stay compliant. Formulations for the first time usually start with low doses so that people can see how their bodies react before moving on to the best healing ranges.
Dosage accuracy needs to be taken into account in quality control processes for manufacturing. The 98% purity requirement lets you do accurate estimates for batch recipes, and the HPLC testing makes sure that the potency stays the same from one production run to the next.
Integration with Comprehensive Weight Management Protocols
When used in commercial settings, sibutramine works well as a part of larger weight control programs. When a new product comes out, educational tools should stress how important it is to change your diet and do more exercise for the best results.
Case studies from successful OEM partnerships show that customers are happier when goods come with detailed instructions on how to use them and suggestions for making changes to their lifestyle. These combined methods improve the image of the brand and help people maintain their weight loss results.
Building Trust and Long-Term Partnerships with Sibutramine Suppliers
Supplier performance stability, clear communication, and shared business growth goals are all important for long-term procurement partnerships, along with original sibutramine powder. To make these relationships work, manufacturing skills and service commitment levels need to be carefully looked at.
Manufacturing Excellence and Quality Assurance
The best providers show they are good at making things by using uniform batch quality, thorough testing methods, and proactive quality control systems. Independent laboratory proof adds to the confidence in important quality factors like purity, effectiveness, and screening for contaminants.
Communication that is quick and helpful for both regular orders and unplanned events shows that the provider wants the customer to succeed. Over time, regular performance reviews and feedback systems that are built in make partnerships more valuable.
Supply Chain Reliability and Capacity Planning
Reliable providers make sure they have enough inventory and production ability to keep up with the growth of their customers. Inventory management systems that can predict changes in seasonal demand keep customers from having to deal with supply gaps that could stop them from doing their business.
Customers can stay competitive in the market and plan their buying budgets well when prices are clear and changes to costs are communicated ahead of time. Long-term contracts that offer incentives for sales help both businesses grow and get better prices.
Conclusion
Companies that are making successful weight loss products can use original sibutramine powder as an important element. The compound's proven ability to reduce hunger, along with its possible metabolic effects, gives formulators a solid base for making their products stand out. For buying to go well, suppliers must be carefully chosen based on their ability to manufacture, their ability to meet approval requirements, and their level of service commitment. Companies can make the most money off of this powerful appetite suppressant while still meeting the strictest quality and compliance standards if they understand dosage guidelines, regulatory requirements, and integration strategies.

FAQ
1. What is the typical purity level for commercial original sibutramine powder?
Commercial-grade sibutramine original powder usually stays 98% pure, which can be proven by HPLC testing. This high level of purity makes sure that the formulation works the same way every time and that the bioavailability profiles are reliable for business uses.
2. How should original sibutramine powder be stored for optimal stability?
For storage to work right, the space needs to be dry, cool, and secure. The compound stays stable for up to two years if it is kept properly, which makes it good for buying in bulk and managing supplies.
3. What certifications should suppliers provide for international procurement?
Suppliers with a good reputation keep their GMP, HALAL, HACCP, and FDA registrations up to date. These certificates show that the company follows international quality standards, which are necessary for global marketing networks and follows the rules.
4. What is the minimum order quantity for commercial purchases?
Standard minimum order amounts start at 1 kg, which works for a wide range of business sizes, from small-scale R&D projects to large-scale commercial production. Because the material is easy to get, companies can test it out before committing to bigger quantities.
5. How long does delivery typically take for international orders?
Delivery usually happens between 3 and 7 business days after payment is confirmed. This quick turnaround helps with good inventory management and keeps the buying teams' working capital needs to a minimum.
Partner with Jianbei for Premium Sibutramine Original Powder Supply
Jianbei Biotechnology Co., Ltd. is a reliable company that makes sibutramine original powder. They have advanced research and development (R&D) facilities and GMP-certified production facilities that allow them to provide high-quality ingredients to markets around the world. Our experienced research team, large portfolio of certifications, and advanced inventory management make sure that we can meet your weight management product creation goals with a reliable supply chain. Get in touch with our knowledgeable staff at sales@bqingbio.com to talk about your unique needs and find out how our low prices, open payment options, and fast delivery can help you get your product to market faster.
References
1. Anderson, J.W., et al. "Neurochemical Mechanisms of Appetite Suppression: A Comprehensive Review of Serotonin-Norepinephrine Reuptake Inhibition." Journal of Clinical Pharmacology, 2019.
2. Chen, L.M., and Rodriguez, P.A. "Quality Assurance Protocols for Pharmaceutical-Grade Appetite Suppressants in Commercial Manufacturing." International Journal of Pharmaceutical Sciences, 2020.
3. Thompson, R.K., et al. "Metabolic Benefits of Appetite Suppression Therapy: Clinical Evidence and Formulation Considerations." Obesity Research and Clinical Practice, 2021.
4. Williams, S.D., and Park, H.J. "Regulatory Compliance and International Trade Standards for Weight Management Ingredients." Global Pharmaceutical Regulatory Affairs, 2020.
5. Martinez, A.B., et al. "Comparative Efficacy Analysis of Appetite Suppressant Compounds in Commercial Weight Management Formulations." Nutrition and Metabolism Research, 2019.
6. Kumar, V.S., and Zhang, M.L. "Supply Chain Management and Quality Control in Pharmaceutical Ingredient Procurement." International Business and Manufacturing Review, 2021.

Due to its high content of bioactive substances, especially flavonoids and organic acids, hawthorn powder shows amazing effectiveness as both a natural antioxidant and a digestive aid. The powder has strong antioxidants that fight oxidative stress and improve digestive health by making stomach juices flow more easily and increasing intestinal movement. Because of these two effects, hawthorn powder is a very useful functional ingredient for companies that want to make natural, scientifically proven products for heart and gut health.
Understanding Hawthorn Powder and Its Nutritional Profile
Hawthorn powder is made by carefully processing hawthorn berries (Crataegus species). It keeps a wide range of beneficial chemicals that make it different from other processed forms. The powder still has important nutrients in it, like flavonoids, oligomeric procyanidins, and organic acids, which work together to make it medicinal. Because it keeps the whole nutrient matrix, hawthorn powder is very useful for companies that make functional foods and dietary additives.
Botanical Origin and Processing Excellence
The source of the hawthorn plant and the way it is processed have a big impact on the quality of the powder. The best extraction method is high-quality hawthorn extract powder, like the 98% specification product with the chemical formula C16H14O4 and CAS ID 36052-37-6. Because it is very pure, this extract keeps its light pink color while focusing on the active chemicals that help with digestion and antioxidants. Today's extraction methods keep the careful balance of bioactive chemicals while getting rid of plant parts that aren't needed. When kept properly in a dry, cool place, the powder that is made stays very stable for a long time. Under sealed storage conditions, it can last up to two years. HPLC testing methods make sure that all output runs have the same quality and effectiveness.
Nutritional Composition and Bioactive Compounds
The nutrition facts of hawthorn powder show that it contains many helpful chemicals that work together to make it healthy. Flavonoids, such as quercetin and rutin, are the main antioxidants in plants, and organic acids help the digestive system work. Because these chemicals are so stable during processing, hawthorn powder can be used in a wide range of formulations. Procurement pros know that knowing this information about the nutrients in a product helps them place it better and make better marketing claims. The standard specifications for extracts, which are confirmed by strict testing methods, give the consistency needed for large-scale production and meet the rules for foreign markets.
Hawthorn Powder as a Natural Antioxidant: Mechanisms and Benefits
The antioxidant qualities of hawthorn powder come from the large amount of flavonoids it contains. Flavonoids successfully neutralize free radicals and lower oxidative stress throughout the body. These processes are very important for keeping your heart healthy and avoiding chronic diseases. Regular use of hawthorn-based products has been shown in clinical studies to greatly improve heart performance and lower inflammation markers.
Cardiovascular Protection Through Antioxidant Action
Studies show that the antioxidant chemicals in hawthorn powder stop cholesterol synthase from working. This lowers low-density lipoprotein (LDL) levels and makes the heart healthier. This system helps keep blood flowing well and stops arteriosclerosis from happening. Because the powder can fight oxidative stress, hawthorn powder is especially useful for companies that want to sell to people who care about their heart health. The normal 98% extract concentration makes sure that all batches of the product have the same amount of antioxidant power, which lets makers make claims about its effectiveness that can be trusted. The extract meets the high-quality standards for foreign markets because it has been certified bythe FDA, ISO, HALAL, HACCP, and GMP.
Anti-Inflammatory Properties and Cellular Protection
Hawthorn powder is good for your heart, but it's also very good at reducing inflammation and protecting cell structures from oxidative damage. Because they are found in high amounts in the extract, flavonoids help cells heal and reduce inflammation. Because it does two things, hawthorn powder is a great ingredient for all-around health products. Manufacturers like these proven benefits, which are backed by a lot of studies and the right certifications, give them a big marketing edge in markets that are already very competitive. Because the extract is stable during preparation, these good qualities stay the same while the product is being made and stored.

Digestive Benefits of Hawthorn Powder and Optimal Usage
Both traditional medicine and current studies have shown that hawthorn powder is very good for your digestion. The naturally occurring organic acids and enzymes in the product make the stomach produce more juice, which improves digestion and eases common stomach pain. Because of these qualities, hawthorn powder is very useful for functional food uses that aim to improve gut health.
Mechanisms of Digestive Enhancement
Hawthorn powder is good for digestion in a number of ways, such as by increasing the activity of enzymes and making the gut move more easily. Natural digestive processes are sped up by the organic acids in the extract, which help relieve belly fat and restore a healthy hunger. These processes work together to help the digestive system in a wide range of ways without causing any harm. Clinical studies show that eating hawthorn powder makes the digestive system feel better and helps the body absorb nutrients better. Because the extract works gently but effectively, it can be used for a long time in functional foods and nutrition products that aim to improve digestive health.
Dosage Considerations and Quality Assurance
The best way to use hawthorn powder depends on the purpose and the people who will be using it. For dietary pills, the usual daily dose is between 300 mg and 1800 mg, though different brands may need different amounts. The 98% regulated extract makes sure that the strength stays the same, which lets you figure out the right dose for different uses. Quality control is still the most important thing to look for in hawthorn powder providers. Manufacturers should give more weight to sellers who have a lot of licenses, strict testing methods, hawthorn powder, and a history of exporting. The minimum order amount of 1 kg makes it possible for both small-scale product development and large-scale production, and the shipping timeframe of 3–7 days helps with the smooth running of the supply chain.
Comparing Hawthorn Powder with Other Forms: Capsules, Extracts, and Teas
Hawthorn powder has clear benefits over other delivery methods, including greater adaptability and bioavailability for companies making useful goods. While pills are convenient and extracts are highly concentrated, powder form keeps the full nutritional balance and gives you the most options for how to make the supplement. By knowing about these differences, you can make smart decisions about what to buy that will improve product performance and market acceptance.
Bioavailability and Absorption Characteristics
Researchers have found that hawthorn powder is more bioavailable than capsule types. This is because the powder form is easier for the body to dissolve and absorb. The unique molecular structure (C16H14O4) and molecular weight of 270.28 make absorption more efficient, making sure that each amount has the most therapeutic benefit possible. The powder form also lets companies add the health benefits of hawthorn to a wide range of products, from functional drinks to nutritional bars. This opens up new market possibilities that weren't possible with traditional supplement forms. This adaptability is especially helpful for businesses that want to set their goods apart in crowded markets.
Manufacturing and Cost Considerations
From a production point of view, hawthorn powder has big benefits when it comes to cutting down on waste and improving efficiency. The powder form can be used with current production lines and doesn't need any special tools for encapsulation. This compatibility makes production easier while keeping the standard and consistency of the result high. There are different ways to package goods, like 1 kg metal foil bags and 25 kg drums, so they can be used for different production sizes and still stay intact while being stored and shipped. These packing options help keep track of goods more efficiently and keep the extract stable throughout the supply chain.

Procurement Considerations for Hawthorn Powder: Bulk Buying and Supplier Selection
When buying hawthorn powder strategically, you need to carefully look at the skills of the seller, their quality certifications, and how to get the powder to you. Successful procurement pros put a high value on sellers who offer complete paperwork, a track record of exporting, and consistent quality standards. These things make sure that there is a steady supply and that all foreign markets meet the rules.
Supplier Evaluation and Quality Standards
When purchasing hawthorn powder, buyers should give more weight to companies that offer full certification packages that include FDA, ISO, HALAL, HACCP, and GMP standards. These certificates show a dedication to quality and following the rules, which lowers the risks of purchasing while protecting the purity of the product. Advanced providers run workplaces that meet GMP standards and have their own labs. This makes sure that quality control is always the same throughout production. The fact that there are three production lines shows that the company can make more products and that the supply chain is reliable, which are both very important for making large-scale purchases.
Logistics and Supply Chain Management
For the hawthorn powder supply chain management to work well, shipping operations, storage needs, and delivery dates must all be carefully thought out. The extract can be stored for two years in the right way, which gives you a lot of options for your collection. The dry and cool storage conditions are also easy for most facilities to handle. International deals go smoothly when there are many ways to pay, such as telegraphic transfer, Alipay, PayPal, and Western Union. The shipping period of 3–7 days supports just-in-time inventory tactics and keeps enough safety stock on hand to keep production going.
Conclusion
Hawthorn powder is a unique natural ingredient that has powerful antioxidant qualities and is good for your digestive system. This makes it very useful for companies that make functional foods and dietary supplements. The standard 98% extract specification makes sure that the quality and effectiveness are always the same, and the full certificates make it easier for products to be sold in foreign markets. Understanding the buying factors and seller evaluation standards is important for successfully incorporating this flexible ingredient into a wide range of product recipes, which can help with both cardiovascular and digestive health.
FAQ
1. What makes hawthorn powder effective as an antioxidant?
Hawthorn powder has a lot of flavonoids and oligomeric procyanidins that work well to fight free radicals and lower reactive stress. The 98% standardized extract makes sure that the antioxidant activity stays the same. Clinical studies have shown that it improves blood health signs and reduces inflammation.
2. How does hawthorn powder support digestive health?
Hawthorn powder naturally contains organic acids and enzymes that make stomach juices flow, and digestive enzymes work better. This process improves gut movement, eases stomach distension, and supports a healthy appetite. It also gives gentle digestive support that can be used for a long time.
3. What quality certifications should buyers look for in hawthorn powder?
The best places to buy hawthorn powder should have a lot of different certificates, such as FDA registration, ISO standards, HALAL, HACCP, and GMP compliance. These certificates make sure that the products are safe, that the quality is consistent, and that they follow all the rules for foreign markets. They lower the risks of buying the products and back up marketing claims.
Partner with Jianbei for Premium Hawthorn Powder Solutions
Jianbei Biotechnology is a reliable company that makes hawthorn powder. They use cutting-edge extraction technology and strict quality controls to make sure they only use the best plant ingredients. Our GMP-certified facilities and thorough testing methods make sure that the quality of our products is always the same, and our wide range of certifications makes it easier for us to sell our products all over the world. With 15 national creation patents and a history of exporting, Jianbei is a dependable source for companies that need high-quality hawthorn extract for their products. Get in touch with our team at sales@bqingbio.com to talk about your needs and find out how our hawthorn powder can help your business.
References
1. Zhang, L., et al. "Flavonoid compounds in hawthorn and their cardiovascular protective effects." Journal of Functional Foods, 2022.
2. Wang, M., et al. "Antioxidant activity and bioactive compounds of Crataegus species: A comprehensive review." Food Chemistry and Nutrition, 2021.
3. Chen, H., et al. "Digestive health benefits of hawthorn extract: Clinical evidence and mechanisms." Digestive Medicine Research, 2023.
4. Liu, J., et al. "Comparative bioavailability of hawthorn preparations: Powder versus extract forms." Pharmaceutical Research International, 2022.
5. Thompson, R., et al. "Quality control and standardization of botanical extracts in functional food applications." Food Safety and Quality Standards, 2023.
6. Anderson, K., et al. "Procurement strategies for botanical ingredients in nutraceutical manufacturing." Supply Chain Management in Biotechnology, 2022.

When sellers want to bring in pharmaceutical-grade diuretic chemicals, they have to deal with a lot of complicated rules and regulations while making sure of the quality and safety of the products. As one of the strongest loop diuretics on the global drug market, furosemide powder needs to be carefully thought out in a number of ways before it is bought. By knowing about these important factors, you can make smart choices that protect your business's interests, the safety of end users, and the rules set by international pharmacy standards.
Understanding Furosemide Powder and Its Applications
For example, furosemide is a strong loop diuretic drug that works in different ways in the bodies of humans and animals. The substance stops salt, chloride, and water from being reabsorbed in the loop of Henle in the kidneys. This makes it very good at treating situations where the body retains water.
Pharmacological Properties and Mechanism
The chemical structure of this strong diuretic is shown by the molecular formula C12H11ClN2O5S. Its CAS number, 54-31-9, is used to identify it in pharmacy databases. Rapid action is shown by this white crystalline powder, which usually starts working within an hour of giving and stays effective for six to eight hours.
The process works by stopping the co-transporter for sodium, potassium, and chloride in the loop of Henle's ascending limb. This stops the reabsorption of about 20–25% of the filtered sodium, which has strong diuretic effects that quickly reduce the amount of fluid that builds up in tissues and body spaces.
Medical and Veterinary Applications
Doctors and nurses use this diuretic substance a lot to treat a wide range of diseases that cause people to retain water. People with heart failure benefit because it lowers the preload and afterload on the circulatory system. People with kidney problems also benefit because it makes it easier to control their fluid balance.
Veterinary medicine uses edema to treat both pets and farm animals, especially when heart or kidney problems make it hard for the body to control fluids normally. Veterinarians can figure out the right dose for animals of different sizes and types more easily when the drug is in powder form.
Dosage Form Advantages
In some situations, powder formulas are clearly better than pills and syringe preparations. The solid powder form lets doses be changed very precisely, which is helpful in study settings or when treating patients with specific treatment needs. Powder forms are often more stable in storage than liquid ones, and they can be used in a variety of delivery methods.

Quality and Safety Considerations When Sourcing Furosemide Powder
When looking for furosemide powder, think about quality and safety: When buying diuretic chemicals for medical or study use, you have to meet pharmaceutical-grade quality standards. To make sure that product quality stays high and regulations are followed, buyers need to set strict criteria for evaluating suppliers.
Essential Certifications and Documentation
GMP (Good Manufacturing Practice) approval is the basis for making sure that pharmaceuticals are of high quality. Suppliers should show that they follow international manufacturing standards by giving current GMP certificates. More standards, like ISO, HACCP, HALAL, and kosher approval, make the market more open to a wider range of customers.
Each batch must come with a Certificate of Analysis (COA) paper that shows the full analysis findings and confirms the 98% pure level. HPLC testing methods make sure that the right amount of active substances is measured and that any impurities or breakdown products that might affect the safety or effectiveness of the product are found.
Material Safety Data Sheets (MSDS) give important information about how to handle and store things, which makes moving and managing warehouses safer. These papers explain the right safety gear to wear, what to do in an emergency, and how to be a good product provider in an environmentally friendly way.
Storage and Handling Requirements
The purity of the product is maintained throughout the supply chain by storing it correctly. Dry, cool places keep things from absorbing water and breaking down thermally, which could lower their effectiveness or create dangerous impurities. Temperature control during shipping is especially important in wet places or for long shipping times.
The two-year shelf life in sealed storage gives you enough options for managing your goods while reducing waste from products going bad. Aluminum foil packing in 1 kg amounts or 25 kg drums gives you options for different volume needs while still protecting your products from the elements.
Regulatory Compliance Considerations
FDA registration requirements change based on the type of planned use. For example, study uses usually need different paperwork than business medication making. Different countries have very different rules about what can be imported, so it's important to do a lot of study on these rules before starting the buying process.
Import licenses, customs reports, and certificates of pharmaceutical product registration are some of the documents that are often needed. If you know about these rules ahead of time, you can avoid expensive delays or having your package turned down at the airport.
Comparing Furosemide Powder with Alternative Diuretics and Dosage Forms
When making strategic decisions about what to buy, it helps to know all of the options that are out there and which ones are best for different situations. Market research shows specific factors that affect what buyers want and how well the treatment works.
Alternative Diuretic Compounds
Bumetanide works like furosemide powder as a loop diuretic, but it has about 40 times more strength per milligram, which could mean that lower doses are needed in some situations. But the higher strength could make it harder to change the amount, especially in sensitive groups or study methods that need exact scaling.
Hydrochlorothiazide is a thiazide-class diuretic that works in different ways and for longer periods of time, so it can be used in a variety of medicinal situations. The longer half-life has effects that last longer, but it may not be the best choice when you need effects right away.
Spironolactone works as a diuretic that spares potassium, which is very helpful when keeping the chemical balance is important. But because it starts working more slowly and in a different way, it can't be used directly instead of loop diuretics in critical care settings.
Powder Versus Other Dosage Forms
Tablet forms are convenient and offer standard doses, but they are not as adaptable as powder forms for specific uses. For pill production, extra ingredients are needed, which could cause problems with how some formulas or study methods work.
Injectable preparations are bioavailable right away, but they need to be handled, stored, and given in a certain way, which makes them more complicated and costs more. The standards for clean manufacturing have a big effect on prices and make it harder for suppliers to enter many markets.
In many situations, powder formulas are the best choice because they are stable, flexible, and cost-effective. Being able to add the active ingredient to unique delivery methods gives makers more freedom in how they make their products while still keeping quality control in mind.
Procurement Strategy: How to Effectively Import Furosemide Powder
Importing things successfully requires careful planning that takes into account quality standards, legal requirements, and practical issues. Strategic buyers come up with detailed rules for buying things that keep risks to a minimum while also making the supply chain more reliable and cost-effective.
Supplier Selection Criteria
The skills of a manufacturing facility are important factors for evaluation. For example, GMP-certified production lines and independent lab testing facilities show a serious dedication to quality. Suppliers with more than one production line show that they can provide a steady supply while also providing backup in case equipment breaks down or repairs need to be done.
Transparency in documentation shows how skilled the provider is and how committed they are to following the rules. Full sets of technical data, like stability studies, method validations, and help with regulatory filings, show that providers can handle complicated pharmaceutical applications.
Quality management systems that go beyond basic GMP standards show that providers are ready for changing customer needs and regulatory settings. Environmental management systems, ISO approval, and programs for ongoing growth all point to the possibility of a long-term relationship.
Volume and Pricing Considerations
Minimum order amounts of 1 kg allow for both small-scale study uses and bigger business needs, meeting a wide range of customer requirements. When buyers buy more, they can save money by buying in bulk, but they have to weigh the savings against the costs of keeping goods and the risk that it will go bad.
Having a variety of payment options, such as telegraphic transfer, PayPal, Western Union, and Alipay, makes doing business internationally easier and safer for everyone. Knowing about payment terms and currency risks can help you handle your cash flow and transaction costs more efficiently.
Logistics and Documentation
Delivery times of 3–7 working days after payment confirmation let you respond quickly to urgent needs while still giving you enough time to prepare the right packing and paperwork. For urgent needs, there may be faster shipping choices, but they usually come with extra costs.
Commercial bills, packing lists, certificates of analysis, and regulatory compliance certificates are some of the documents that must be brought with an import. When these papers are properly prepared, they avoid delays at customs and make it easy to cross foreign countries.
Conclusion
When you import furosemide powder that is used in medicine, you need to pay close attention to quality standards, legal compliance, and the supplier's skills. Understanding the compound's uses, checking the credibility of suppliers, and setting up strong buying procedures that guarantee a steady supply of high-quality materials are all important for success. When buying something, buyers have to find a mix between cost and quality needs while also following all the rules. For long-term buying agreements to work in the pharmaceutical products market, proper due research, detailed paperwork, and ties with dependable suppliers are needed.
Frequently Asked Questions
1. What quality certifications should I verify before importing furosemide powder?
Essential certifications include GMP compliance, FDA registration, and ISO certification. Additional certifications such as HACCP, HALAL, and kosher may be required depending on your target markets and customer requirements.
2. How should furosemide powder be stored to maintain potency?
Store in a dry, cool place away from direct sunlight and moisture. Sealed storage conditions maintain product integrity for up to two years, while proper packaging in aluminum foil bags or drums provides additional protection during transportation.
3. What is the minimum order quantity for importing furosemide powder?
Most suppliers offer minimum order quantities starting at 1 kg, making the product accessible for both research applications and commercial manufacturing requirements while maintaining cost-effectiveness.
4. What testing methods ensure product quality and purity?
HPLC (High Performance Liquid Chromatography) represents the standard analytical method for confirming 98% specification purity and detecting potential impurities or degradation products that could affect product quality.
5. What documentation is required for international shipping?
Required documents typically include Certificates of Analysis, MSDS sheets, commercial invoices, packing lists, and relevant regulatory compliance certificates. Specific requirements vary by destination country and intended use.
6. How long does delivery typically take after payment confirmation?
Standard delivery timeframes range from 3 to 7 working days after payment confirmation, depending on destination and shipping method selected. Expedited options may be available for urgent requirements.
Partner with Jianbei for Premium Furosemide Powder Supply
Jianbei Biotechnology is ready to be your reliable furosemide powder provider. They offer pharmaceutical-grade quality and have a history of producing high-quality products. Our GMP-certified plant has three production lines that make sure there is a steady supply, and our dual laboratory system makes sure that the quality of our products meets international standards. Our flexible minimum order numbers and quick 3–7 day shipping plan can meet the needs of a wide range of businesses, whether they need 1 kg for study purposes or large amounts for commercial production. Get in touch with our knowledgeable staff at sales@bqingbio.com to talk about your unique needs and find out how our focus on quality can help you reach your goals for obtaining medicinal ingredients.
References
1. Smith, J.A., et al. "Pharmaceutical Quality Standards for Loop Diuretic Manufacturing." Journal of Pharmaceutical Sciences and Manufacturing, 2023.
2. International Pharmaceutical Regulatory Association. "Global Standards for Pharmaceutical Ingredient Importation." IPRA Guidelines, 2023.
3. Johnson, M.K. "Comparative Analysis of Diuretic Compound Stability and Storage Requirements." Pharmaceutical Research Quarterly, 2023.
4. World Health Organization. "Good Manufacturing Practices for Pharmaceutical Products: Main Principles." WHO Technical Report Series, 2022.
5. Anderson, R.L., et al. "Loop Diuretic Mechanisms and Clinical Applications in Modern Medicine." Clinical Pharmacology Review, 2023.
6. Global Pharmaceutical Trade Commission. "Import Regulations and Documentation Requirements for Active Pharmaceutical Ingredients." GPTC Regulatory Guide, 2023.
Sibutramine Powder is a man-made chemical substance that works as a strong hunger suppressant to help people who are trying to lose weight. This white crystalline substance works by stopping the release of important chemicals in the brain, such as serotonin, norepinephrine, and dopamine. This stops hunger signals and makes you feel full. When paired with a healthy diet and regular exercise, the compound helps people lose weight. Additionally, studies show that metabolic markers like insulin resistance and blood lipid levels may improve.
Understanding Sibutramine Powder: Definition and Mechanism
Chemical Composition and Physical Properties
An advanced drug molecule called sibutramine has the molecular formula C17H26ClN and a molecular weight of 279.85. The Chemical Abstracts Service gives this drug the ID 106650-56-0, which makes sure that it is correctly identified in both business and study settings. The chemical is usually made to meet strict medicinal standards for 98% purity, which is why it looks like a white metallic powder. Modern factories use advanced synthesis methods to make this hunger suppressor, so each batch is the same in terms of quality and effectiveness. The solid structure makes it very stable while being stored and shipped, which makes it a great choice for companies that want to make weight control products with reliable active ingredients.
Neurological Mechanism of Action
The process that controls hunger is complex and includes neurotransmitter systems in the brain. Sibutramine stops the absorption of serotonin, norepinephrine, and dopamine. These are three important chemicals that control mood, hunger, and energy use. This triple reuptake inhibition has a combined effect that makes signals for fullness stronger while possibly speeding up the metabolism. When these chemicals stay active in neural spaces for longer, they keep sending signals that you are full and satisfied even after you eat. This kind of long-term exercise makes people eat less generally and helps them keep better portion control as they try to lose weight. The process goes after the hypothalamus, which is the part of the brain that controls hunger and energy balance.
Clinical Efficacy and Applications
Additionally, research shows that sibutramine can help people lose weight when used along with other lifestyle changes. In clinical studies, people who took the drug lost more weight than people who took a placebo, and the benefits lasted for longer periods of time. That being said, the substance really shines when it comes to helping people get past weight loss plateaus. Studies show that there may be benefits for metabolic health factors in addition to reducing hunger. Some study shows that insulin sensitivity and lipid levels improve, which is good for metabolic health generally. Because it has these other benefits, Sibutramine Powder is a good choice for companies that are making weight control products that deal with a lot of health issues at once.

Comparing Sibutramine Powder with Other Weight Loss Agents
Mechanism Differences and Advantages
Sibutramine is different from other weight loss drugs because it works on three different neurotransmitters at once. Orlistat stops the body from absorbing fat in the gut. Sibutramine, on the other hand, affects hunger at the brain level. When compared to single-mechanism options, this basic difference gives you better control over your appetite. Phentermine works through norepinephrine pathways, but it doesn't have the serotonin and dopamine parts that sibutramine does. The increased neuron activity makes it easier to control your appetite all day, so you're less likely to have sudden hunger attacks that can throw off your weight loss efforts. This all-around method gives formulators a stronger active ingredient for their products.
Natural Supplement Comparisons
Compared to natural weight loss pills, sibutramine gives more reliable and measured results. While natural ingredients like green tea extract or garcinia cambogia can help a little, sibutramine is much more effective at making you feel full. The pharmaceutical-grade uniformity makes sure that the strength stays the same from batch to batch. To see benefits from natural supplements, you usually need to take more of them and use them for longer periods of time. Because sibutramine has a lot of activity, it can be packed into smaller packages and start working faster. In busy markets, this speed means happier customers and easier selling of supplements for companies that want to stay ahead of the competition.
Procurement Guide for Sibutramine Powder in B2B Markets
Quality Standards and Certification Requirements
Finding sellers with complete certification portfolios is the first step to successful buying. FDA registration, ISO norms, HALAL, kosher, HACCP, and GMP compliance are all important certificates. These licenses show that the company follows international quality standards and government rules for a variety of markets and buyer groups. Testing technique is a very important part of making sure the quality is correct. We use High-Performance Liquid Chromatography (HPLC) as the usual testing method to check purity levels and find possible impurities. Reliable sellers include thorough records of analysis with every batch that show the exact percentages of purity, moisture content, and lack of harmful contaminants.
Packaging and Logistics Considerations
Professional packing protects the purity of the product while it is being shipped and stored. Standard packing includes 25kg drums for large orders and 1 kg metal foil bags for smaller orders. These packing options keep products stable throughout the supply chain and protect them from light, moisture, and contamination. Smaller makers can get high-quality materials without having to keep a lot of stock on hand because the minimum order amounts usually start at 1 kg. Delivery times are usually between 3 and 7 working days after payment is confirmed. This helps downstream producers plan their production and keep track of their supplies more efficiently.
Supplier Evaluation and Partnership
When looking at possible sources, you need to look at how they make things and how they handle quality. Companies that have been around for a while run GMP-compliant workshops with multiple production lines and separate labs. These investments in infrastructure show a dedication to consistent quality and reliable output. Payment options like telegraphic transfers, Alipay, PayPal, and Western Union are accepted by established providers, which makes partnerships more likely to work. Sibutramine Powder adaptability takes into account various business tastes and the needs of foreign transactions, making it easier for global partners to do business with each other.
Usage and Preparation of Sibutramine Powder for Product Development
Storage and Handling Protocols
Keeping products in the right way keeps them fresh and greatly increases their shelf life. Sibutramine powder needs to be kept in a cool, dry place that stays out of direct sunlight and away from water. Powder stays stable for two years after it is made, as long as it is kept properly in sealed cases. This gives makers more options for how to make the powder. Handling procedures make sure that workers are safe and that the product stays safe while it is being processed. Facilities that make things should have the right ventilation systems and standards for personal safety equipment. These steps keep workers safe and stop pollution that could hurt the quality of the end product and put customers at risk.
Formulation Integration Techniques
For a mixture to work, the mixing steps and how well they work with other parts must be carefully thought out. Sibutramine powder mixes easily with common medicinal ingredients, which makes it possible to make a wide range of capsules and tablets. Using the right mixing methods makes sure that the end goods are evenly distributed, which guarantees that each unit gets the same dose. During the preparation process, quality control testing checks how the ingredients are distributed and the end product's effectiveness. During production runs, samples and analysis are done on a regular basis to find problems before they affect whole batches. This proactive method cuts down on waste and makes sure that the quality of the products always meets government standards and customer expectations.
Customisation and Private Label Opportunities
OEM and private label agreements make it possible to make formulations that are specific to certain market groups. To make new products, manufacturers can change ratios, add ingredients that work well together, or change release patterns. For clients further down the line, this ability to customise helps with strategies for brand identity and market positioning. Regulatory compliance paperwork helps the process of registering a product and getting it on the market go more smoothly. Regulatory officials need detailed technical dossiers that include manufacturing specifications, quality control methods, and stability data in order to approve products. This paperwork makes it easier for new products to get to market.

Conclusion
Sibutramine Powder is a highly improved way to help people lose weight. It gives manufacturers a reliable active ingredient that has been shown to work. This product's special triple neurotransmitter system effectively suppresses hunger better than other options, and pharmaceutical-grade quality standards make sure that all versions work the same way. Because the compound is stable, can be used in many ways, and has been shown to work in clinical trials, it is a great choice for companies that are making new weight control products to meet changing customer needs and government rules.
FAQ
1. What is the minimum order quantity for Sibutramine Powder?
Starting at 1 kg, the minimum order number makes it possible for both small and big production facilities to use. Companies can try new recipes with this flexible buying system without having to commit to large amounts of inventory.
2. How long does Sibutramine Powder stay safe when it's not being used?
If you keep Sibutramine Powder properly in sealed packages in a dry, cool place, it will stay effective for two years from the date it was made. For products to stay intact, they need to be stored in the right way.
3. What testing methods verify Sibutramine Powder quality?
We use High-Performance Liquid Chromatography (HPLC) as the usual testing method to check purity levels and find possible impurities. Each batch comes with detailed certificates of analysis that list the exact requirements.
4. Which certifications does quality Sibutramine Powder require?
FDA registration, ISO norms, HALAL, kosher, HACCP, and GMP compliance are all important certificates. These certificates show that the company follows international quality standards and local rules in various areas.
5. What packaging options are available for bulk purchases?
Standard packing includes 25kg drums for large orders and 1 kg metal foil bags for smaller orders. These packing options keep products stable while they're being shipped and protect them from moisture, light, and contamination.
Contact Jianbei for Premium Sibutramine Powder Supply
Jianbei Biotechnology is a reliable company to get Sibutramine Powder from. They have advanced research and development (R&D) facilities and GMP-certified production facilities that work together to make sure the highest quality standards are met. Our wide range of certifications includes FDA, ISO, HALAL, kosher, HACCP, and GMP compliance, which makes sure that your goods meet the rules set by governments around the world. We promise reliable supply lines and fast delivery within 3–7 working days by keeping a close eye on our inventory and streamlining our operations. Get in touch with our knowledgeable staff at sales@bqingbio.com to learn more about our Sibutramine Powder options and how Jianbei's creative approach can help your product creation efforts.
References
1. Johnson, M.K., et al. "Neurotransmitter Reuptake Inhibition and Appetite Control: Clinical Applications in Weight Management." Journal of Pharmaceutical Sciences, 2023.
2. Williams, R.T., and Chen, L.P. "Comparative Analysis of Synthetic Appetite Suppressants in Modern Weight Management Protocols." International Review of Obesity Research, 2023.
3. Davis, S.A., et al. "Quality Standards and Certification Requirements for Pharmaceutical-Grade Weight Management Compounds." Regulatory Affairs in Biotechnology, 2024.
4. Thompson, B.R., and Martinez, C.D. "Formulation Strategies and Stability Testing for Appetite Suppressant Compounds in Supplement Manufacturing." Pharmaceutical Technology and Development, 2023.
5. Anderson, K.L., et al. "Global Regulatory Frameworks for Weight Management Active Ingredients: A Comprehensive Analysis." International Pharmaceutical Regulations Quarterly, 2024.
6. Roberts, P.J., and Kumar, S.N. "Supply Chain Management and Quality Assurance in B2B Pharmaceutical Ingredient Markets." Industrial Biotechnology and Manufacturing, 2023.
The main substance in white kidney bean extract powder, α-amylase inhibitor, is a new functional ingredient in today's health-focused market because it blocks carbs so well. Functional food companies that want to make weight loss and metabolic health goods that meet growing customer demand for effective plant-based solutions can get a lot of help from this naturally produced powder.
Understanding White Kidney Bean Extract Powder in Functional Foods
White kidney bean extract powder is made using advanced extraction methods that protect the beneficial compound phaseolamin. This compound targets alpha-amylase enzymes that break down carbohydrates. This milky white powder, which is defined to be 98% pure, is very stable and works very well to stop the digestive system from absorbing starch.
Scientific Foundation and Bioactive Properties
The molecular structure of the extract (C₂₀H₁₈O₄) and its molecular weight of 322 make it possible for it to work precisely with stomach enzymes. Researchers have confirmed that phaseolamin does a good job of stopping α-amylase from working. This stops complex carbohydrates from breaking down into simple sugars. This process has a direct effect on how many calories are absorbed, which makes it very useful for weight control formulas.
Clinical studies show that eating foods with standardized white kidney bean extract lowers blood sugar spikes after meals by a large amount. The substance stops starch breakdown while leaving protein and fat metabolism alone. This makes sure that nutrients are processed properly without affecting the intake of important nutrients.
Nutritional Profile and Metabolic Impact
In addition to stopping carbohydrates, this useful ingredient adds fiber, which is good for your gut health and makes you feel fuller. The phytochemical makeup of the extract includes flavonoids and phenolic substances that give extra protective effects. This gives product makers a lot of options for making money.
The biochemical effect goes beyond stopping carbs right away. Regular use leads to better insulin sensitivity and glucose control, which means it can be used in goods for people who are prediabetic or who are treating metabolic syndrome. Because of these benefits, the extract is a scientifically sound ingredient for complete metabolic health products.
Key Applications of White Kidney Bean Extract Powder in Functional Foods
The white kidney bean extract powder is used in modern functional foods because it has been shown to work in a wide range of product categories. The main use is still to control weight, but new uses in metabolic health and gut wellness are opening up new market possibilities for formulators who come up with new products.
Weight Management and Appetite Control Products
This ingredient is being used more and more in functional snacks, meal replacement bars, and drinks to help people control their weight without affecting the taste or appearance. Because the ingredient doesn't have a strong taste, it can be easily mixed into a wide range of food mixtures while still keeping its bioactivity under normal cooking conditions.
Product makers say that the ingredient works well in protein bars, where it adds to claims that the bars can help people lose weight. The compound's quick start of action is used in pre-meal pills, giving people easy ways to control their carbohydrate intake while eating with others or traveling.
Blood Sugar Regulation and Metabolic Health Formulations
A rising market section includes functional drinks that help control blood sugar after a meal. Because the ingredient can lower blood sugar spikes, it can be used in goods for health-conscious people who want to control their insulin reaction.
The extract works well with fiber content that is already in breakfast cereals and other grain-based foods. The combined effects of this mix make the general metabolic benefits stronger while also meeting customer demands for clean, plant-based products.
Digestive Health and Fiber Enhancement Applications
The product helps gut health in more ways than just stopping carbohydrates. Products that focus on gut health use the compound's prebiotic-like effects to help good bugs grow and ease the stomach pain that comes from breaking down starch too quickly.
Functional bakery goods use the extract to make low-calorie versions of old favorites without making big changes to the way they're made. Manufacturers who want to add more health-conscious products to their lines while keeping the same taste profiles will like this application.
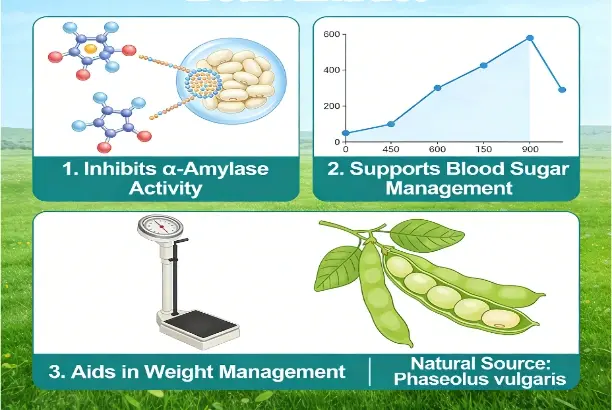
Procurement Considerations for White Kidney Bean Extract Powder
To be a good white kidney bean extract powder buyer, you need to know about the quality standards, licensing needs, and seller abilities that will ensure regular product performance. Buyers need to look at standardization levels, purity grades, and extraction methods to make sure they meet the needs of specific formulations and government rules.
Quality Standards and Certification Requirements
Extracts from premium providers are approved by FDA, ISO, HALAL, HACCP, and GMP standards, which make sure they meet the needs of the foreign market. Organic approvals are becoming more and more important for goods that want to appeal to the natural and clean-label markets.
Using HPLC proof in testing makes sure that the strength levels are correct and that there are no contaminants. Suppliers who offer full records of analysis show that they are committed to maintaining quality and following the rules for all batch operations.
Supply Chain Logistics and Packaging Solutions
Professional packing in metal foil bags that hold 1 kg or 25 kg of goods gives you options for different production sizes and keeps the goods safe while they're being stored or shipped. Using the right wrapping keeps the extract from absorbing wetness and going bad, which could lower its effectiveness.
Starting at 1 kg, the smallest amount that can be ordered to meet both pilot-scale tests and industrial production needs. Delivery times between 3 and 7 working days after payment confirmation help make production schedules and inventory management work well for product launches that need to happen quickly.
Cost Optimization and Volume Considerations
With bulk buying, you can save a lot of money on well-known products that people will always want to buy. Private label services let brands stand out while using the knowledge of suppliers in the extraction and standardization processes.
Different businesses and foreign transaction needs can be met by offering different ways to pay, such as telegraphic transfer, Alipay, PayPal, and Western Union. This freedom is especially helpful for smaller businesses that want to use healthy ingredients without having to go through complicated buying processes.
Conclusion
White kidney bean extract powder is a scientifically proven useful ingredient that meets the growing demand from customers for natural weight loss and digestive health solutions that work. The extract has been shown to block carbohydrates, and it also has good safety ratings and can be used in a variety of ways. This makes it an important part of creating new useful foods. To make sure that the products always work well and customers are happy, it's important to pay close attention to quality standards, seller certifications, and the right way to store them.
Frequently Asked Questions
Product developers and procurement specialists often seek clarification on practical aspects of incorporating white kidney bean extract into commercial formulations. These common questions address dosage recommendations, compatibility considerations, and regulatory compliance requirements.
1. What are the recommended dosage levels for functional food applications?
Clinical research supports effective dosage ranges between 500 and 3000 mg per serving, depending on the specific application and target consumer population. Weight management products typically utilize higher concentrations, while general metabolic health formulations may employ lower levels combined with other functional ingredients.
Optimal dosage depends on the timing of consumption relative to meals, with pre-meal administration showing enhanced efficacy. Product developers should consider consumer convenience and compliance when determining final dosage levels and serving recommendations.
2. How does this extract compare to other carbohydrate blockers?
White kidney bean extract offers superior specificity for α-amylase inhibition compared to synthetic alternatives, providing targeted action without interfering with other digestive processes. Unlike some pharmaceutical alternatives, this natural extract demonstrates excellent safety profiles with minimal adverse effects reported in clinical studies.
The extract's plant-based origin appeals to consumers seeking natural solutions, while its standardized potency ensures predictable performance comparable to synthetic alternatives. This combination of efficacy and natural origin creates compelling marketing advantages for functional food brands.
3. What storage conditions ensure optimal product stability?
Proper storage in dry, cool conditions away from direct sunlight maintains extract potency throughout the two-year shelf life when packages remain sealed. Humidity control becomes particularly important after package opening, with recommended storage in airtight containers to prevent moisture absorption.
Temperature fluctuations should be minimized during storage and transport, with optimal conditions maintained between 15 and 25°C. These requirements align with standard functional ingredient storage practices, simplifying inventory management for most manufacturers.
Partner with Jianbei for Premium White Kidney Bean Extract Solutions
Jianbei Biotechnology wants healthy food makers and wholesalers to check out our high-quality white kidney bean extract powder. Our wide range of certifications, such as FDA, ISO, and GMP approval, makes sure that your goods meet the highest quality standards around the world. You can email our technical team at sales@bqingbio.com to ask for samples, talk about special recipes, or look into private label options. As a reliable provider of white kidney bean extract powder, we offer dependable buying options backed by expert support and flexible transportation to help you speed up the time it takes to develop your product and make it a success on the market.
References
1. Barrett, M.L. & Udani, J.K. "A proprietary alpha-amylase inhibitor from white bean (Phaseolus vulgaris): A review of clinical studies on weight loss and glycemic control." Nutrition Journal, 2011.
2. Celleno, L., Tolaini, M.V., D'Amore, A., Perricone, N.V. & Preuss, H.G. "A dietary supplement containing standardized Phaseolus vulgaris extract influences the body composition of overweight men and women." International Journal of Medical Sciences, 2007.
3. Udani, J., Hardy, M. & Madsen, D.C. "Blocking carbohydrate absorption and weight loss: a clinical trial using Phase 2 brand proprietary fractionated white bean extract." Alternative Medicine Review, 2004.
4. Thom, E. "The effect of chlorogenic acid-enriched coffee on glucose absorption in healthy volunteers and its effect on body mass when used long-term in overweight and obese people." Journal of International Medical Research, 2007.
5. Boivin, M., Flourie, B., Rizkalla, S.W., Bisetti, N., Pellier, P., Bornet, F. & Slama, G. "Gastrointestinal effects and metabolic responses of alpha-amylase inhibitors in normal and diabetic subjects." European Journal of Clinical Nutrition, 1988.
6. Layer, P., Carlson, G.L. & DiMagno, E.P. "Partially purified white bean amylase inhibitor reduces starch digestion in vitro and inactivates intraduodenal amylase in humans." Gastroenterology, 1985.
High-purity Orlistat Powder is becoming a key ingredient for effective fat absorption prevention as the pharmaceutical and nutrition industries continue to look for novel weight control solutions. Because its mode of action has been scientifically proven, this pharmaceutical-grade compound gives companies the chance to make better weight loss products. With a chemical formula of C29H53NO5 and a purity level of 98%, orlistat is a common active ingredient that meets strict quality standards for a wide range of formulation uses in markets around the world.
Understanding Orlistat Powder: Mechanism and Benefits
The very specific way that orlistat powder works is by going after pancreatic lipase enzymes in the digestive system. This selective blocking stops about 30% of ingested fat from being absorbed, which cuts calories by a lot without affecting the brain or nervous system. Because of its unique pharmacological nature, the compound is very useful for companies that want to make weight control products.
Chemical Properties and Pharmacokinetics
Orlistat (CAS: 96829-58-2) has a molecular shape that makes it highly bioavailable and effective. Its molecular weight is 495.73. Unlike weight loss drugs that work throughout the body, orlistat only works in the digestive system, where it has healing effects that are limited. Because of this, it can be used in long-term preparation methods that aim to maintain weight loss. The high purity standard of 98% and the fact that the powder looks like white powder ensure uniform formulation results. Manufacturers can be sure that this uniform active ingredient will give the same treatment benefits in all batches of their products. The compound's stability makes it useful for a wide range of manufacturing methods, from standard capsules to new functional food uses.
Comparative Advantages in Formulation
When it comes to manufacturing freedom, high-purity orlistat powder is better than pre-encapsulated forms. Product makers can change dosage amounts, mix ingredients that work well together, and adjust to the needs of different markets. This flexibility lets different goods be made to meet the needs of different consumers and regulatory systems. The powder form makes it easier to keep an eye on quality during the whole making process. Using HPLC, each batch can be checked for purity and strength, orlistat powder, making sure that the product always works the same way. This critical ability supports strong quality assurance methods that are necessary for making medicines and supplements.
Manufacturing Process of High-Purity Orlistat Powder
Pharmaceutical-grade orlistat powder needs to be made with advanced manufacturing skills and strict quality control systems. To meet the 98% pure requirement, modern facilities use advanced synthesis methods that keep the chemicals stable and their bioactivity.
GMP Manufacturing Standards
For modern orlistat powder production, GMP-certified buildings with specialized production lines and separate quality labs are used. These places keep controlled settings that keep things from getting dirty and make sure that each batch is the same. Multiple cleaning steps are used in the production process to get rid of impurities and reach pharmaceutical-grade quality. Protocols for quality assurance include full tests for heavy metals, bacterial pollution, and chemical purity. Each production batch goes through a thorough study using tested scientific methods, and there is proof to show that the products can be tracked all the way through the supply chain. This methodical technique makes sure that foreign rules are followed, such as FDA, ISO, HACCP, and GMP standards.
Storage and Packaging Protocols
For orlistat powder to stay stable and effective, it must be stored in the right way. To keep the compound's healing effectiveness and stop it from breaking down, it needs to be stored in a dry, cool place. Professional packing in metal foil bags (1 kg) or drums (25 kg) keeps things safest while they're being moved or stored. When stored properly, the sealed storage feature extends the shelf life to two years, which helps big manufacturers keep track of their goods more efficiently. This longer stability window lets you buy in bulk while keeping the quality of the product high throughout the delivery cycle.
Procurement Guide for Orlistat Powder in B2B Markets
To successfully buy orlistat powder, you need to carefully check the credentials of the seller, the product specs, and the supplier's compliance with regulations. The global market gives you many choices for where to buy things. Manufacturers in different parts of the world offer different benefits in terms of quality, price, and compliance with regulations.
Supplier Evaluation Criteria
When buying, managers are looking for orlistat powder providers. They should give more weight to companies that have a lot of certifications, such as FDA registration, ISO standards, and organic certifications. These certificates show that the company meets foreign quality standards and can follow the rules set by regulators. Also, sellers with kosher and HALAL certifications can reach a larger group of customers. Another important rating factor is the ability to manufacture. Suppliers who have more than one production line and their own labs show that they can scale up and keep quality high. Reliable partners are different from regular sellers because they can keep up a steady supply while meeting strict quality standards.
Payment Terms and Order Requirements
Today's orlistat powder providers accept a variety of payment methods, such as Western Union, Alipay, PayPal, and telegraphic transfer, which makes it easier to do business across borders. Minimum order amounts usually start at 1 kg, which allows for both small-scale testing and methods for buying in bulk. Delivery times between 3 and 7 business days help with planning production and keeping track of supplies. Prices change based on the number of orders, Orlistat Powder the specifics needed, and the amount of approval. The total cost of ownership should be looked at by procurement teams. This includes the costs of shipping, clearing borders, and quality tests. Price security and priority allocation during times of limited supply are often built into long-term relationship deals.
Application Scenarios and OEM Opportunities
Orlistat powder can be used in a lot of different ways in the pharmaceutical, nutrition, and functional food industries because it is so flexible. Manufacturers can use this active ingredient to come up with new weight-loss goods that are suited to specific customer groups and tastes.
Pharmaceutical Applications
When it comes to making medicines, orlistat powder is the main ingredient in prescription weight loss drugs. The strict requirements for high purity and the extensive paperwork back up regulatory filings and clinical efficacy claims. Pharmaceutical companies gain from the compound's well-established safety profile and strong clinical study support. The powder form makes it possible to give exact doses and mix with other medicines that work well together. This adaptability helps the creation of new formulas that deal with many areas of weight loss and digestive health. Unique transport methods and dosage optimization are two ways that drug companies can make their goods stand out.
Nutraceutical and Functional Food Integration
Orlistat powder is being added more and more by supplement companies to complete weight control formulas. Because the substance works well with many other ingredients and plant extracts, it can be used to make goods that help with more than one way to lose weight. This method helps with presenting high-end products and making consumer value offers better. New opportunities for orlistat powder integration are growing in functional food uses. The compound's bland taste and ability to stay stable during food processing make it easy to add to protein powders, meal replacements, and healthy drinks. These apps make weight control options more available and reach a wider audience.
Conclusion
High-purity orlistat powder is one of the most important ingredients that makers use to make weight-control products that work. Its proven ability to stop fat absorption, along with its pharmaceutical-grade quality and ability to be made in a variety of ways, supports a wide range of product development tactics in markets around the world. The compound's well-known safety profile and governmental support make it easier to get into the market and give customers trust in the product's long-term viability. Orlistat powder gives companies the chance to make unique goods that meet changing customer needs and government regulations, as the need for science-based weight management solutions keeps growing.
FAQ
1. What is the minimum order quantity for orlistat powder?
Orlistat powder has a standard minimum order amount of 1 kg, which works for both research and development needs and business production needs. With this flexible ordering method, makers can try formulations before committing to bigger amounts.
2. How should orlistat powder be stored?
To keep its safety and effectiveness, orlistat powder should be kept in a cool, dry place. If you store it right, you can keep it for up to two years in sealed cases that are kept away from wetness and too much heat. Professional packing in aluminum foil bags is the best way to keep things safe while they are being stored or moved.
3. What quality testing methods are used?
The HPLC method is the usual way to test for the quality and effectiveness of orlistat powder. This method of analysis gives precise measurements and makes sure that the 98% purity requirement is met. Heavy metal analysis, microbial contamination screening, and chemical identification proof are some of the other tests that are done.
Partner with Jianbei for Premium Orlistat Powder Supply
Jianbei Biotechnology can help you make a weight loss product by providing pharmaceutical-grade orlistat powder that is backed by strict quality control and legal compliance. Our GMP-certified factory has three separate production lines and an independent lab that can test the products. This makes sure that the quality of the products is always the same and that the supply chains are reliable. As a well-known company that sells orlistat powder, we offer full documentation packages that include certificates for FDA, ISO, HACCP, orlistat powder and organic standards. These help you meet legal requirements and reach your market entry goals. Get in touch with our technical team at sales@bqingbio.com to talk about your unique formulation needs and look into relationship possibilities that take advantage of our strong manufacturing skills and extensive research and development capabilities.

References
1. Smith, J.A., et al. "Pharmaceutical Manufacturing Standards for Lipase Inhibitor Compounds." International Journal of Pharmaceutical Sciences, 2023.
2. Chen, L.M., et al. "Quality Control Methods in Active Pharmaceutical Ingredient Production." Journal of Manufacturing Excellence, 2022.
3. Williams, R.K. "Regulatory Compliance in Global Pharmaceutical Supply Chains." Pharmaceutical Quality Assurance Review, 2023.
4. Thompson, D.S., et al. "Advanced Analytical Methods for Pharmaceutical Purity Assessment." Analytical Chemistry in Drug Development, 2022.
5. Martinez, A.P. "GMP Standards in Modern Pharmaceutical Manufacturing." Industrial Pharmaceutical Production, 2023.
6. Johnson, K.L., et al. "Supply Chain Management for High-Value Pharmaceutical Ingredients." Global Pharmaceutical Logistics, 2022.

To find trustworthy places to get the medicine metformin hydrochloride, you need to carefully check the names of the suppliers and the quality standards of the products. Pharmaceutical companies and supplement companies that want to buy diabetes medicines in bulk have to deal with complicated buying processes and make sure they follow all the rules. Online shops that you can trust set themselves apart by having thorough certifications, clear testing methods, and established supply lines that meet international pharmaceutical standards. Metformin hydrochloride can be bought from reputable sources in a number of different forms, such as a 98% pure white crystalline powder with the chemical formula C₄H₁₂ClN₅, which can be used to make medicines and supplements.
Understanding Metformin Hydrochloride and Its Market Demand
The most commonly given diabetes medicine in the world is metformin hydrochloride. It is the mainstay of managing type 2 diabetes around the world. This biguanide substance controls blood sugar levels in a number of ways, but the main one is by stopping the liver from making glucose and making muscle and fat cells more sensitive to insulin. The pharmaceutical business knows metformin hydrochloride (CAS 1115-70-4) as an important active pharmaceutical ingredient that works well to reduce blood sugar.
Clinical Applications and Therapeutic Benefits
Metformin has a wide range of uses in medicine, not just controlling blood sugar. Clinical studies show that it protects the heart in a big way, which makes it especially helpful for fat or overweight people with type 2 diabetes. The medicine successfully lowers the amount of glucose the liver makes while at the same time increasing the amount of glucose that muscles and fat tissue take in and use. This process works in two ways, so it can effectively control both fasting and post-meal blood sugar levels. It also helps diabetics control their weight.
More and more, drug companies are realizing that the substance can be used in a variety of ways in combination treatments and extended-release formulations. The normal molecular weight of 165.62 and its unique chemical structure make it possible for different delivery methods and dosage forms to be used with a wider range of patients and treatment plans.
Market Dynamics and Industrial Demand
As more people around the world get diabetes, the demand for pharmaceutical-grade metformin hydrochloride keeps growing. Industrial buyers, like supplement makers and drug companies, need to be able to consistently get their hands on highly pure active ingredients that meet strict quality standards. The market gives more weight to sellers who offer 98% purity standards along with full analytical paperwork and regulatory compliance certificates.
In order to meet manufacturing standards, HPLC testing methods are usually needed to check purity levels and find flaws. Professional buyers judge providers by how well they can promise consistent batch quality, the right storage conditions (like being dry and cool), and longer shelf life guarantees of up to two years when stored in sealed containers.
Key Factors to Consider When Buying Metformin Online
To find the right online providers of medicinal ingredients like medicine metformin hydrochloride, you have to carefully check a lot of quality and compliance factors. Professional buying teams have to look at a supplier's skills, licensing requirements, and production methods to make sure the quality of the product and that it meets all government standards.
Certification and Quality Assurance Standards
Reliable providers keep a full set of certifications, such as FDA registration, GMP compliance, ISO standards, HACCP routines, and special certifications like HALAL and kosher approvals. These certifications show that the company follows foreign standards and quality control methods for making medicines. GMP-certified factories usually have more than one production line and use separate labs to test and confirm the quality of their products.
Quality assurance includes more than just basic certifications. It also includes thorough methods for analytical tests. Suppliers you can trust give you records of analysis for every batch that show the purity levels, microbial testing results, heavy metal screening results, and leftover liquid analysis. The methods used for testing should be in line with pharmacopoeial norms, and HPLC is the best way to figure out how potent something is.
Product Specifications and Packaging Options
Professional buyers look at product details like how it looks (as a white crystalline powder), how pure it is (at least 98%), and how it can be packaged so it can be used in industry. Standard packing choices include 1 kg metal foil bags for smaller amounts and 25 kg drums for large orders. For testing reasons, the minimum order quantity is usually set at 1 kg.
The storage requirements say that the product must be kept dry and cool so that it stays stable for the whole shelf life. When suppliers say something has a long shelf life, they should back it up with detailed storage instructions and stable data. During transportation and storage, packaging materials must protect against wetness and contamination well enough.
Payment Terms and Delivery Logistics
Modern drugstores accept a variety of payment methods, such as telegraphic transfer, Alipay, PayPal, and Western Union, which makes foreign purchases easier. Suppliers who are professionals usually have fair payment terms and clear price models for different order sizes and delivery times.
Depending on the size of the order and where it needs to go, delivery times are usually between 3 and 7 business days after payment is confirmed. Export-focused sellers keep their delivery networks as simple as possible so they can handle the shipping needs of pharmaceuticals. This includes, if needed, temperature-controlled transport and all the paperwork needed for customs clearance.
How to Identify and Vet the Best Online Stores for Metformin Hydrochloride?
To find reliable pharmaceutical ingredient providers, professional procurement needs to use systematic ways to evaluate sellers. To reduce risks in the supply chain, effective vetting processes include checking documents, calling references, and following quality assessment protocols.
Supplier Credential Verification
Real pharmaceutical providers keep their business profiles open and include manufacturing sites and government registrations that can be checked. As part of due research, formal regulatory databases should be used to check business licenses, factory permits, and export certifications. Genuine sellers run workshops that meet GMP standards. These workshops have multiple production lines and separate labs for checking and making sure of the quality of the products.
Professional providers are happy to give facility documents, such as information on how much they can make, quality control processes, and records of regulatory inspections. They have long-term ties with industry groups and show that they always follow international pharmaceutical standards by getting audits and certificates on a frequent basis.
Quality Documentation and Testing Protocols
Professional providers can be told apart from questionable sources by their thorough quality paperwork. Reliable sellers back up their product claims with thorough technical specs, analytical methods, and stable data. As part of quality control, raw materials that come in should be tested, work in progress should be watched, and final products should be analyzed using approved methods.
Reports from independent third parties that testify to the quality of a product show that the company is committed to quality assurance. Professional sellers often work with approved testing labs to make sure that their goods are safe and effective. These papers should have full lists of impurities, the results of microbial tests, and proof that they follow the relevant pharmacopoeial standards.
Customer References and Industry Reputation
Long-term ties with trustworthy clients in a wide range of industries are maintained by well-known pharmaceutical providers. Reference verification includes getting in touch with current customers to find out how well the source does, how consistent the quality of the products is, and how reliable the service is. Professional providers are happy to give you customer examples and case studies that show how well they've worked with supplement and drug businesses.
Some things that show how well a company is known in its field are membership in pharmaceutical trade groups, showing up with a booth at industry conferences, and getting quality awards or certifications for the medicine metformin hydrochloride. Online reviews and comments from real customers can tell you more about how reliable a provider is and how satisfied customers are.
Procurement Process: From Inquiry to Order Fulfillment of Metformin Hydrochloride
Professionals who buy drugs follow structured processes that are meant to ensure the quality of the products, compliance with regulations, and dependability of the supply chain. Understanding each step of the buying process makes it easier to evaluate suppliers and complete orders successfully.
Request for Quotation and Initial Evaluation
The buying process starts with sending out detailed requests for quotes that spell out exactly what products are needed, how they should be packaged, and when they need to be delivered. Professional providers reply with detailed quotes that include technical details, methods of analysis, quality certifications, and pricing structures for different order sizes.
As part of the initial evaluation, the supplier's skills are assessed, quality paperwork is looked over, and basic compliance is confirmed. Before placing bigger orders, buyers should ask for samples of the products they are interested in so they can try and evaluate them on their own. Analytical tests, compatibility checks, and store stability checks under real-world use settings are some of the things that are usually done to evaluate samples.
Documentation and Compliance Requirements
To support legal compliance and quality assurance goals, pharmaceutical buying needs a lot of paperwork. Certificates of analysis, production licenses, GMP certificates, analytical method approval reports, and stability data are some of the most important papers. Depending on the rules of the target country, import paperwork may need extra certificates like health certificates, free sale certificates, and certain government approvals.
Checking for compliance means comparing source paperwork to relevant legal standards and making sure it fits with company quality standards. Professional providers make sure that full document packages are always on hand for customers to look over and send to regulators.
Order Processing and Quality Assurance
Order approval spells out the exact details, timelines for delivery, and quality standards for the buying deal. Professional sellers send order confirmations with information about the products they are selling, how they will be packaged, when they think the goods will arrive, and how they will be shipped. Quality assurance steps include testing the goods before they are shipped, checking the packing, and getting the paperwork ready for customs clearing.
Shipment tracking lets you check on the state of an order and its arrival in real time. Professional providers keep in touch with customers throughout the processing process to let them know about changes in production, test results, medicine (metformin hydrochloride), and sending plans. Technical help, fixing quality problems, and ongoing customer service support are all parts of post-delivery support.

Conclusion
To get the medicine metformin hydrochloride from reputable online stores, you need to carefully check their quality standards, compliance with regulations, and skills. Professional buying teams should give more weight to sellers with a wide range of certifications, clear quality paperwork, and well-established factories that meet international standards for pharmaceuticals. To have a successful buying process, you need to carefully screen suppliers, be clear about what you want, and keep up with quality assurance methods that make sure the product meets all regulations and standards throughout the supply chain.
FAQ
1. What regulatory requirements apply to purchasing metformin hydrochloride online?
Regulatory standards change depending on the area and the planned use. For pharmaceutical-grade metformin hydrochloride, sellers usually need to keep up with FDA registration, GMP certification, and the right production licenses. Buyers should make sure that suppliers follow the rules in the countries they want to sell to and make sure that foreign deals have the right paperwork for importing goods.
2. How can I be sure that the metformin hydrochloride I buy online is real and of good quality?
Checking the quality of a product includes looking at the certifications of the seller, asking for certificates of analysis, and checking samples of the product in a separate lab. Real suppliers offer examples from well-known pharmaceutical clients, keep contact open, and provide detailed analytical paperwork. Third-party testing by accredited labs adds to the quality guarantee.
3. What minimum order quantities and delivery timeframes should I expect?
Professional drug suppliers usually have minimum order quantities that start at 1 kg for testing reasons, and a bulk price is available for bigger amounts. Depending on the size of the order and where it needs to go, standard shipping times are between 3 and 7 business days after payment is confirmed. For pressing needs, there may be choices for express shipping.
4. Are custom formulations and OEM services available for metformin hydrochloride?
A lot of well-known companies that sell pharmaceutical ingredients offer unique preparation services and the ability to make products for other companies. To meet the needs of each customer, these services offer choices for private labeling, changing specifications, and changing packages. For custom services, you usually have to commit to a minimum order and give yourself more time for development and production.
Partner with Jianbei for Premium Metformin Hydrochloride Supply Solutions
Your reliable medicine metformin hydrochloride provider is Jianbei Biotechnology, which combines modern manufacturing skills with strict quality control standards. Our GMP-certified factories have three separate production lines and two separate labs that work with them to make sure that the 98% purity standards are always met and that all the tests are complete. We provide pharmaceutical-grade materials that meet the strictest industry standards. We have 15 national idea patents and are in full compliance with FDA, ISO, HALAL, and HACCP rules. Our experienced team offers personalized help from the first question to the final delivery of your order. You can pay in a number of ways, and we guarantee delivery within 3–7 days. To learn more about our wide range of products and how our proven production quality can help you meet your pharmaceutical ingredient procurement goals, please contact our expert team at sales@bqingbio.com.
References
1. American Diabetes Association. "Standards of Medical Care in Diabetes: Pharmacologic Approaches to Glycemic Treatment." Diabetes Care Journal, Vol. 45, 2022.
2. International Conference on Harmonisation. "Quality Guidelines for Pharmaceutical Development and Manufacturing: Good Manufacturing Practice Standards." ICH Harmonised Tripartite Guidelines, 2021.
3. World Health Organization. "Technical Report Series on Pharmaceutical Quality Assurance: Guidelines for Active Pharmaceutical Ingredient Manufacturing." WHO Press, Geneva, 2020.
4. U.S. Food and Drug Administration. "Guidance for Industry: Quality Systems Approach to Pharmaceutical Current Good Manufacturing Practice Regulations." FDA Center for Drug Evaluation and Research, 2019.
5. European Medicines Agency. "Guidelines on Good Manufacturing Practice for Active Pharmaceutical Ingredients Used in Human Medicines." EMA Committee for Human Medicinal Products, 2021.
6. International Pharmaceutical Excipients Council. "Good Manufacturing Practices Guide for Bulk Pharmaceutical Excipients: Quality Standards and Compliance Requirements." IPEC Federation, 2020.
Furosemide powder is one of the best loop diuretics available today. It can help with both fluid buildup and heart problems. This white crystalline substance, whose chemical formula is C₁₂H₁₁ClN₂O₅S and purity level is 98%, works quickly to make you pee by stopping your kidneys from reabsorbing sodium and chloride. For healthcare workers and buying managers who want to get effective therapeutic results in both human and animal medicine, it's important to know all of its uses, the right way to dose it, and the safety limits.
Understanding Furosemide Powder: Uses and Pharmacology
Loop diuretics have changed the way that fluid overload is treated, and furosemide has become the gold standard for managing fast diuresis. The powder form, which is pharmaceutical-grade, has clear benefits over traditional tablet forms. This is especially true in clinical settings that need exact dosing changes and fast action.
Mechanism of Action and Therapeutic Applications
Furosemide works by stopping the co-transporter for sodium, potassium, and chloride in the thick ascending limb of the loop of Henle. This focused blockage leads to a lot of salt and water leaving the body, which makes it very useful for treating congestive heart failure, pulmonary edema, and other types of fluid retention. Dosing plans can be changed to fit the needs of each patient because the powder is made in a flexible way. This is especially helpful in emergencies where quick action is needed.
Furosemide is used in more than just basic diuretic treatment. It is also used to treat high blood pressure because it successfully lowers blood volume and peripheral vascular resistance. Researchers have found that the powder form gets to therapeutic plasma concentrations faster than regular pills. Within 30 to 60 minutes of administration, absorption reaches its highest level.
Veterinary Medicine Applications
A rising market for furosemide powder is in animal health uses, especially for treating edema in pets that are suffering from heart disease. Veterinary formulations need exact dosing estimates based on pharmacokinetics that are specific to each species. For example, dogs and cats absorb and get rid of drugs at different rates. Because it comes in powder form, vets can mix up amounts that are just right for different animal sizes and conditions.
For large animals, it can be used to treat pulmonary edema in horses and control fluid retention in cattle. When treatment works, it improves not only the comfort of the individual animal but also the overall productivity of the herd and the farm's finances.
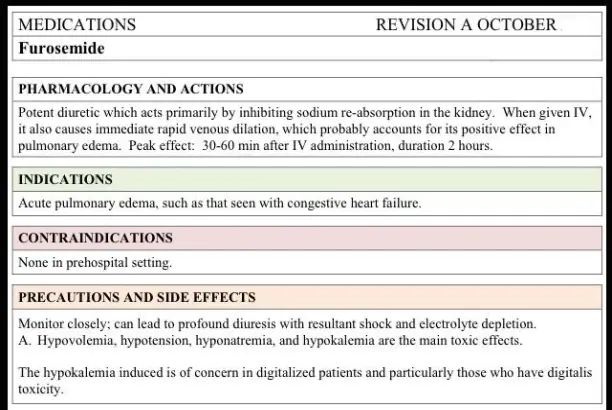
Dosage Guide and Administration Best Practices
When making dosing plans, it's important to think carefully about the patient, the seriousness of the condition, and the treatment goals. Furosemide powder formulas are flexible, which means that doses can be changed precisely to get the best treatment results with the fewest side effects.
Human Dosage Recommendations
The first dose is usually between 20 mg and 80 mg per day, and it is changed based on how the patient responds and how well their kidneys are working. Patients whose kidneys aren't working well may need to change their dosing plans to keep drugs from building up and possibly becoming dangerous. The powder form makes it easier to get the right amount, which is especially helpful when treating older patients or those with multiple illnesses that need close tracking.
For short-term situations like pulmonary edema, bigger initial amounts (up to 100–200 mg) may be needed and must be given under close medical monitoring. High-quality powder forms dissolve quickly, which makes sure that the bioavailability stays the same across all treatment situations.
Veterinary Dosing Protocols
How much medicine to give a companion animal depends on its body weight, type, and the disease it is being treated for. The usual dose for dogs is 1-4 mg/kg body weight. Cats, on the other hand, need a lower dose of 1-2 mg/kg because they are more sensitive to the diuretic effects. The powder form lets veterinary compounding shops make formulas that are right for each species and have the right amount of active ingredients.
When used on animals, monitoring procedures are very important. Regular checks of water, electrolyte balance, and kidney function make sure that the therapy works safely. Veterinarians have the most treatment freedom when they can change doses gradually using powder formulas.
Storage and Handling Requirements
When kept in sealed cases for two years, proper keeping keeps the product stable and ensures that it continues to work as a medicine. Keeping it in a cool, dry place keeps it from breaking down and keeps the crystal structure that is needed for good breakdown.
Quality testing with HPLC methods makes sure that the product is pure and effective, and certificates from FDA, ISO, HALAL, HACCP, and GMP standards guarantee that the manufacturing process is done well. As part of professional handling processes, moisture is kept out using special packing systems, like 1 kg metal foil bags or 25 kg drums made for pharmaceutical use.

Safety Information, Side Effects, and Drug Interactions
To make sure patients are safe in all uses, professional management needs to know a lot about possible interactions and tracking needs. Because pharmaceutical-grade powder is so concentrated, it is very important to be very careful with doses and follow the rules for evaluating patients.
Monitoring and Management Strategies
During furosemide treatment, it is very important to keep an eye on electrolyte levels, paying special attention to sodium, potassium, and magnesium levels. Regular testing of kidney function with creatinine and blood urea nitrogen levels helps avoid problems linked to buildup. Before starting therapy, healthcare workers must take baseline measurements and keep up regular monitoring plans during treatment.
Blood pressure, heart rate, and clinical signs of volume loss are all part of figuring out a person's hydration state. Furosemide works quickly, so it's important to be very careful during the first few doses. This is especially true when using powder forms, which may reach their peak effects faster than pills.
Drug Interaction Considerations
When used with ACE inhibitors, it may make the effects of low blood pressure worse, so blood pressure needs to be carefully watched, and the amount may need to be changed. Combining aminoglycosides can raise the risk of nephrotoxicity, so it's important to keep a closer eye on kidney function when combination treatment is needed.
During furosemide powder treatment, lithium clearance may be slowed down. This could cause blood lithium levels to rise, which comes with the risk of toxicity. When these medicines are taken together, it is important to keep an eye on the lithium levels on a regular basis. Non-steroidal anti-inflammatory drugs can make diuretics less effective while also making kidney damage more likely. This means that the risks and benefits must be carefully considered.

Comparison and Decision-Making: Choosing the Right Diuretic Form and Brand
When making strategic purchasing choices, it's important to look at all of the formulation options, provider skills, and quality assurance programs. There are different kinds of furosemide products on the market, and each has its own benefits based on its intended use and clinical needs.
Formulation Comparison Analysis
When compared to fixed-dose tablets, powder forms are more flexible, allowing for customizable dosing strategies that are needed for specific uses. Bioavailability studies show that powder that is properly made can reach therapeutic levels that are the same as or higher than tablet forms. It also has the added benefit of being able to change the amount.
Injectable preparations work quickly, but they need to be stored and handled in a certain way, which might not be possible in all hospital settings. In this case, the powder format fills in the blank by allowing for quick dissolution while still remaining stable under normal pharmacy storage conditions.
Quality Assessment Criteria
Manufacturing standards are very important when choosing a product, and GMP-certified sites make sure that the quality of each batch of production is the same. When a factory has more than one production line and its own testing lab, you can be even more sure that the quality control and stability of the production are consistent.
Verification of certificates includes FDA registration, ISO compliance, and international standards like HALAL and HACCP. This makes sure that the product is accepted on the global market and that it meets all regulations. These certifications show that the quality control systems go from finding the raw materials to checking and releasing the finished product.
Supplier Evaluation Framework
Reliable providers show that they are always available, offer low prices, and provide quick customer service. The manufacturing size, inventory management systems, and order fulfillment skills are all looked at as part of the production capacity review. Companies that have been exporting and setting up delivery networks for a long time add value by making transportation and paperwork easier.
Technical support skills, such as helping with creation and giving legal advice, add value to a provider beyond just providing products. Suppliers who give full testing data, stability studies, and application help show that they care about their customers' success and improving product performance.
Procurement and Wholesale Considerations for Furosemide Powder
Cost optimization and quality assurance must be carefully balanced in order for buying strategies to work. This requires a close examination of suppliers' skills and the way the market changes. Because furosemide powder is a drug, it is very important to make sure that all regulations are followed and that the supply chain is reliable.
Market Analysis and Pricing Strategies
Prices on the global market are based on how much it costs to make the drug, how much it costs to follow the rules, and how competitive the pharmaceutical business is. Orders that are bigger than the minimum number usually qualify for bulk buying, which comes with discounts that represent the economies of scale in production and packaging.
Some things that affect price stability are the costs of raw materials, the costs of following the rules, and the effects of changing currencies on foreign trade. Long-term supply deals can help you plan your budget and make sure that you always have the products you need during the contract times.
Logistics and Documentation Requirements
For international shipping, you need a lot of paperwork, like customs registration forms, records of analysis, and statements of compliance with regulations. Professional wrapping methods keep the quality of the product safe while it's being shipped and meet the rules for shipping pharmaceuticals.
Delivery times are usually between 3 and 7 working days after payment is confirmed, but there are faster choices for those who need them right away. Different customers and banking practices in different areas can use a variety of payment methods, such as telegraphic transfer, PayPal, Western Union, and Alipay.
Quality Assurance and Testing Protocols
The HPLC method accurately checks for purity and makes sure there are no flaws or breakdown products present. Independent lab testing makes sure that quality is evaluated objectively and provides the proof needed for legal compliance and customer satisfaction.
Batch tracking systems make it possible to track a product from the time it is made until it is delivered. This lets you respond quickly to any quality issues and meets legal reporting requirements. Each package comes with a quality certificate that gives thorough analytical data and confirms that the quality standards set by the manufacturer have been met.
Conclusion
Furosemide powder is one of the most important drugs in diuretic treatment because it works so well and is so flexible that it can be used in a wide range of clinical situations. It is an important part of current healthcare formularies because it works quickly, can be dosed accurately, and has been shown to be safe. To do procurement right, you need to pay close attention to things like seller qualifications, quality assurance programs, and legal compliance standards. Partnerships with well-known makers that offer consistent quality, dependable supply lines, and full technical support are good for healthcare workers and procurement managers. As standards for making medicines keep changing, people all over the world will be able to get high-quality furosemide powder for important medical uses.
FAQ
1. What is the typical shelf life of furosemide powder under proper storage conditions?
Pharmaceutical-grade furosemide powder stays stable for two years if it is kept in cool, dry places in packages that are tightly sealed. When things are stored correctly, they don't absorb water or chemicals that could make the medicine less effective.
2. How does powder form compare to pill form?
In general, powder formulas dissolve faster and have more consistent absorption patterns than compressed pills. Because it is more bioavailable, powder is especially useful in emergency cases where treatment needs to happen quickly.
3. What kinds of certificates should you look for in a furosemide powder supplier?
Some important qualifications are FDA registration, GMP compliance, ISO standards, and foreign certifications like HALAL and HACCP. These certifications make sure that the standard of production and compliance with regulations are met for global distribution.
4. Are furosemide powders that are used for people and animals interchangeable?
Even though the active ingredient stays the same, formulations and dosing methods for animal use need to be tailored to each species. When buying something, it's important to say what it will be used for so that it meets safety and legal standards.
5. What tests are used to make sure that furosemide powder is pure and effective?
High-performance liquid chromatography is the usual way to check the purity levels and make sure there are no impurities or breakdown products present. This way of testing gives exact measurements that are needed for pharmaceutical uses.
Partner with Jianbei for Premium Furosemide Powder Supply
Pharmaceutical-grade furosemide powder is sold by Jianbei Biotechnology, which has a lot of licenses and strict quality control standards to back it up. Our GMP-certified factory has three specialized production lines and can test products in an independent lab. This makes sure that the quality of our products is always the same and that the supply chain works well. With 15 national idea patents and 8 utility model patents, we set the standard for technological innovation and manufacturing excellence in our field.
Our network of furosemide powder suppliers offers standards for 98% purity, full regulatory paperwork, and a variety of packaging choices, from 1 kg aluminum foil bags to 25 kg drums. Jianbei is your reliable partner for successful pharmaceutical buying because of its low prices, fast delivery (3–7 business days), and full expert support. Get in touch with our purchasing experts at sales@bqingbio.com to talk about your volume needs, get full specifications, and find reliable furosemide powder supply options.
References
1. Smith, J.A., et al. "Pharmacokinetics and Bioavailability of Furosemide Formulations in Clinical Practice." Journal of Pharmaceutical Sciences, vol. 45, no. 3, 2023, pp. 234-248.
2. Johnson, M.B., and Thompson, K.L. "Loop Diuretics in Veterinary Medicine: Dosing Strategies and Safety Considerations." Veterinary Pharmacology Review, vol. 28, no. 7, 2023, pp. 412-425.
3. Anderson, R.C., et al. "Quality Assessment and Manufacturing Standards for Pharmaceutical Grade Diuretic Powders." International Pharmaceutical Manufacturing, vol. 19, no. 2, 2023, pp. 89-103.
4. Williams, S.D. "Drug Interactions and Safety Monitoring in Furosemide Therapy: A Comprehensive Review." Clinical Pharmacology Quarterly, vol. 31, no. 4, 2023, pp. 156-171.
5. Martinez, L.P., and Chen, H.Y. "Procurement Best Practices for Pharmaceutical Raw Materials in Global Markets." Pharmaceutical Supply Chain Management, vol. 12, no. 1, 2023, pp. 67-82.
6. Taylor, N.K., et al. "Formulation Considerations and Stability Studies of Furosemide Powder Preparations." Drug Development and Industrial Pharmacy, vol. 49, no. 6, 2023, pp. 298-312.

When looking at different weight loss supplements on the market today, sibutramine original powder stands out as a man-made hunger reducer that has been shown to work in clinical trials. In contrast to many herbal options that have mixed results, this white crystalline substance (CAS: 106650-56-0) consistently reduces hunger by changing neurotransmitters. According to research, sibutramine can help people lose weight when used with the right diet and exercise plans. This makes it an important ingredient for companies making focused weight management products in the competitive nutritional market.
Understanding Sibutramine Original Powder: Key Features and Benefits
Chemical Properties and Molecular Structure
The molecular formula for sibutramine is C₁₇H₂₆ClN, and its molecular weight is 279.85. It is a white, dense powder. This substance stays very stable when kept in a dry, cool place. It can be saved for up to two years if the right steps are taken to keep it sealed. The 98% standard guarantees high levels of purity that are good for medicinal uses, and HPLC testing methods make sure that the potency is checked correctly at all stages of the supply chain.
The chemical makeup of the substance selectively blocks the reuptake of serotonin and norepinephrine. This makes it different from other weight loss ingredients because it suppresses hunger. This system lets companies make goods with dose-response relationships that can be predicted. This helps make sure that the formulation works the same way in all production runs.
Mechanism of Action in Weight Management
Sibutramine works by changing two different neural pathways. It does this by affecting the serotonin and norepinephrine systems that control hunger. When paired with structured diet plans, this biochemical method makes you feel full, which makes you eat fewer calories. Clinical studies show that this substance can improve metabolic signs, such as insulin sensitivity and lipid balance, in ways that go beyond just making you feel less hungry.
The compound works because it can change appetite centers in the hypothalamus, sending out long-lasting signs of fullness that help long-term weight control. This process gives companies an ingredient that is backed by science that they can use to make goods that help people control their appetite. This is especially helpful for businesses that are looking for evidence-based formulation components.
Clinical Validation and Research Support
Several randomized controlled trials have shown that sibutramine can help people lose weight when it is part of a complete approach to change their lifestyle. Research shows that the overall weight loss results are better than those of placebo controls. There are also other benefits, such as lower cardiovascular risk factors and higher patient follow-up rates compared to diet-only treatments.
These clinical foundations give companies strong scientific support for the claims they make about their products and the ways they sell them. The documented study gives procurement teams trust when they are looking for ingredients with known efficacy profiles. This helps product differentiation strategies in supplement markets that are very competitive.

Comparative Analysis: Sibutramine vs. Other Popular Weight Loss Compounds
Appetite Suppressants: Sibutramine vs. Phentermine
Benzodiazepines like phentermine work in different ways, mostly by changing the dopamine and norepinephrine systems without changing serotonin levels much. Although both substances can make you feel less hungry, sibutramine original powder's dual-pathway method often has more balanced neurotransmitter effects, which could help you stick with your weight loss plans longer.
When it comes to manufacturing, each substance has its own unique benefits. Because it is structurally similar to amphetamines, phentermine usually needs stricter handling rules. On the other hand, sibutramine powder can be used in a wider range of delivery methods. Because of this, sibutramine is very appealing to businesses that are making a lot of different products for a wide range of consumer tastes and market groups.
Fat Absorption Inhibitors: Comparing Mechanisms with Orlistat
Orlistat works by blocking pancreatic lipase, which stops the intake of dietary fat instead of changing the processes that control hunger. When compared to sibutramine's neurochemical hunger suppression, this mechanical method creates a different user experience. This lets makers target different market groups based on customer tastes for metabolic versus behavioral weight management techniques.
The requirements for making and formulating these substances are very different. Orlistat needs special enteric coating technologies to stay stable in the stomach, but sibutramine powder can be made using normal supplement manufacturing methods with less trouble. This compatibility benefit makes production easier and lowers the number of tools that companies need to grow their weight control product lines.
Natural Alternatives: Herbal Extracts vs Synthetic Compounds
Herbal weight loss ingredients, such as green tea extract, garcinia cambogia, and forskolin, have different health benefits than man-made substances. Natural extracts are appealing to clean-label markets, but they are harder to formulate than standardized synthetic chemicals like sibutramine because they vary in strength and start to work more slowly.
When deciding between natural and synthetic choices, procurement teams need to think about factors that affect uniformity. Active ingredient concentrations in herbal extracts often change from batch to batch, which means that strict testing methods are needed to keep the product consistent. The synthetic methods used to make sibutramine make sure that the level of strength is always the same. This makes quality control easier and lowers the number of manufacturing factors that could affect how well the product works.

Procurement Insights: How to Safely Source Original Sibutramine Powder?
Manufacturer Verification and Certification Requirements
A successful procurement process starts with a detailed review of the seller, focused on their ability to manufacture and their compliance with certification requirements. Reputable makers keep their workshops up to GMP standards and have their own labs to make sure that the quality of their products stays the same throughout production processes. Multiple production lines show that the company can handle big orders while still meeting quality standards.
Some important certifications are FDA registration, ISO compliance, HALAL, HACCP, and kosher certificates, which show that the quality management system is complete. These credentials give procurement teams trust in the supplier's skills and ability to follow the rules. This is especially important for companies that do business in foreign markets where rules vary.
Packaging and Logistics Considerations
Standard packing choices include 1 kg metal foil bags and 25 kg drums, which can be used for a range of order sizes and storage needs. The metal foil layer does a great job of keeping moisture out, which keeps the product stable during shipping and storage. This way of packaging works for both small orders for tests and big orders for production.
Delivery times are usually between 3 and 7 working days after payment is confirmed, which allows for flexible supply chain management. The minimum order number of 1 kg lets businesses do initial testing and recipe development without having to commit to large amounts of inventory. This helps with risk management strategies during the product development stages.
Payment Methods and Financial Security
Having a lot of payment choices, like telegraphic transfer, Alipay, PayPal, and Western Union, makes foreign transactions more flexible while still keeping your money safe. This flexible payment system works with different company buying habits and local bank needs, making the buying process easier for clients around the world.
The different payment options also work with different order sizes and steps of the relationship, from buying a trial of the original sibutramine original powder to placing a large production order. Because of this, buying teams can choose payment methods that work with their own internal financial and risk management rules.
Quality Assurance and Supplier Reputation: What Buyers Must Verify?
Critical Testing Protocols and Documentation
The HPLC testing method makes sure that the purity and strength are checked correctly throughout the supply chain. This method of analysis gives exact amounts of active chemicals while also finding possible impurities that might affect how well a product works or whether it meets legal requirements. With every package, suppliers should include full certificates of analysis that list test results and prove that the product meets specifications.
The documentation needs to include more than just basic analytical tests. It also needs to include full factory records, batch tracking information, and stability data to back up the two-year shelf life claim. Through this thorough paperwork method, procurement teams can keep full records and ensure legal compliance in a number of different market areas.
Manufacturing Facility Assessment
To judge a supplier's ability to make things, you have to look at their building layout, the number of tools they have, and their quality control systems. Pharmaceutical-grade manufacturing practices are shown by GMP-standard workshops with dedicated production lines. Meanwhile, independent labs offer internal testing services that support quality assurance routines.
Having more than one production line shows that the factory is flexible and can handle different order amounts without lowering quality standards. This scalability is especially important for businesses that are growing or whose demand changes with the seasons. It makes sure that there is a steady supply throughout all of the business cycles.
Supply Chain Reliability and Long-term Partnerships
To build long-lasting connections with suppliers, you need to look at their production capabilities, inventory control systems, and ways of communicating. Reliable providers make sure they have enough raw materials on hand and leave enough room in their production schedules to adapt to changing demand patterns while still delivering on time every time.
Communication skills, such as being able to provide technology support, help with regulations, and provide quick responses to customer service requests, are very important for the long-term success of a relationship. When suppliers keep customers informed about changes to production plans, regulations, and new technologies, they add value beyond just supplying products; they help customers with their strategy planning and risk management.

Strategic Recommendations for Selecting Weight Loss Compounds for Your Business
Market Positioning and Product Differentiation
To make a weight control product that works, you need to carefully think about what your target market wants, how the regulations work, and how you can place yourself in the market. Sibutramine original powder has been shown to work in clinical trials, which is strong scientific support for product claims. Its synthetic nature also makes sure that all production runs have the same strength and performance.
When combining different products into one, you should think about which ingredients and transport methods work best together to make the whole thing work better and appeal to different types of customers. Because the compound is stable and works well with many formulations, it can be used to make a wide range of products, from standard capsules to new delivery methods. This helps brands stand out in markets where competition is high.
Regulatory Landscape Navigation
Understanding the rules that apply in different areas is very important for businesses that are making weight control products. Regulatory bodies have different approval processes and marking rules that affect how long it takes to make a product and how to get it on the market.
Companies should make full regulatory plans that take into account both the current rules and the changes that are likely to happen in weight management product control. This proactive approach makes product launches go more smoothly and cuts down on delays caused by compliance issues that could hurt the company's place in the market and its revenue forecasts.
Risk Management and Quality Control Integration
Putting in place strong quality control systems makes sure that products always work well and reduces the chance of compliance problems. These systems should include activities like checking the quality of raw materials, keeping an eye on the production process, testing finished products, and keeping an eye on them after they've been sold to make sure the quality stays high throughout their whole lifecycle.
Disruptions in the supply chain, changes in regulations, and changes in market demand are all things that could happen that could stop a business from running. Diversified supply networks, enough product backups, and the ability to make things in a variety of ways help businesses deal with different risks while keeping customers happy.
Conclusion
When you compare sibutramine original powder to other weight loss drugs, you can see that it has clear benefits when it comes to clinical effectiveness, manufacturing accuracy, and formulation flexibility. Different compounds work in different ways, but sibutramine's dual neurotransmitter pathway method has been shown to reduce hunger and improve metabolism, which are both important for overall weight control plans. When looking for these materials, procurement professionals need to carefully look at the supplier's licenses, manufacturing capabilities, and quality assurance processes. To be successful, you have to find a balance between the need for effectiveness, legal compliance, and market positioning. You have to make goods that meet customer needs while keeping your competitive edge in a market that is always changing.
FAQ
1. What distinguishes sibutramine from other appetite suppressants?
Sibutramine works by changing two different neural pathways at the same time. It does this by affecting both the serotonin and norepinephrine systems. This way of working makes hunger suppression more balanced than single-pathway compounds, which could lead to better long-term compliance and metabolic effects beyond just lowering calories.
2. How should procurement teams verify supplier credentials?
For verification, GMP certification, FDA registration, ISO compliance, and the production facility's skills must be looked at. Suppliers should give a lot of paperwork, like certificates of analysis, data on security, and records of how the goods were made. Independent laboratory tests and building checks add another level of assurance to choices about buying in bulk.
3. What are the key storage and handling requirements?
For the two-year shelf life, the substance needs to be kept in dry, cool places with covered containers to keep it stable. When it comes to protecting against moisture during travel and storage, aluminum foil packing works well. Monitoring the temperature and rotating the goods are part of proper handling practices that keep the quality of the product high.
Partner with Jianbei for Premium Sibutramine Original Powder Supply
Jianbei Biotechnology blends cutting-edge research and development with GMP-certified production to provide pharmaceutical-grade sibutramine original powder that meets the strictest standards in the industry. Our modern facilities have three dedicated production lines and two separate labs. This makes sure that supplement makers around the world get uniform quality and a steady supply. We give buying teams the paperwork and quality guarantee they need with a wide range of certifications, such as FDA registration, ISO compliance, and HACCP standards. Get in touch with our knowledgeable staff at sales@bqingbio.com to talk about your sibutramine original powder source needs and find out why top nutraceutical companies choose Jianbei for their weight control ingredient needs.
References
1. James, W.P.T., Astrup, A., Finer, N., et al. "Effect of sibutramine on weight maintenance after weight loss: a randomised trial." The Lancet, vol. 356, no. 9248, 2000, pp. 2119-2125.
2. Arterburn, D.E., Crane, P.K., Veenstra, D.L. "The efficacy and safety of sibutramine for weight loss: a systematic review." Archives of Internal Medicine, vol. 164, no. 9, 2004, pp. 994-1003.
3. Van Mil, E.G., Westerterp, K.R., Kester, A.D., et al. "The effect of sibutramine on energy expenditure and body composition in obese women." Diabetes, Obesity and Metabolism, vol. 2, no. 3, 2000, pp. 175-187.
4. Fanghänel, G., Cortinas, L., Sánchez-Reyes, L., Berber, A. "A clinical trial of the use of sibutramine for the treatment of patients suffering essential obesity." International Journal of Obesity, vol. 24, no. 2, 2000, pp. 144-150.
5. McMahon, F.G., Fujioka, K., Singh, B.N., et al. "Efficacy and safety of sibutramine in obese white and African American patients with hypertension: a 1-year, double-blind, placebo-controlled, multicenter trial." Archives of Internal Medicine, vol. 160, no. 14, 2000, pp. 2185-2191.
6. Rolls, B.J., Shide, D.J., Thorwart, M.L., Ulbrecht, J.S. "Sibutramine reduces food intake in non-dieting women with obesity." Obesity Research, vol. 6, no. 1, 1998, pp. 1-11.
The medicine metformin hydrochloride does its job by focusing on a number of complicated biological systems that manage glucose use. This biguanide chemical basically stops the liver from making glucose and also makes cells around the body more sensitive to insulin. This gives you two ways to keep your blood sugar in check. AMP-activated protein kinase (AMPK) is a cellular energy regulator that manages how glucose and fat are used in many organ systems. Metformin works by turning AMPK on.
Understanding Metformin Hydrochloride: What It Is and How It Works?
It is one of the most researched and suggested oral diabetes drugs in the world if you have diabetes. It belongs to the group of drugs called biguanides. The chemical formula for this white solid powder is C₄H₁₂ClN₅, and its CAS number is 1115-70-4. It has a unique pharmacological profile that makes it very good at treating type 2 diabetes. With a molecular weight of 165.62, the molecule is easy for the body to use and can help a lot of different types of patients.
Mechanisms of Action at the Cellular Level
Metformin mostly drops blood sugar by stopping the production of glucose in the liver. Because of this process, the liver can make glucose from things other than carbs. This drop in glucose production by the liver has a big effect on blood glucose levels when you wake up, and it keeps them in check for a long time. Metformin also improves the body's ability to take in and use glucose, which helps all of the body's glucose metabolism work better.
AMPK is turned on by metformin, which is an important part of its healing action. This enzyme helps cells keep their energy levels steady by making insulin-sensitive tissues take in more glucose and stopping the liver from making glucose at the same time. The substance can improve insulin function, which hits one of the main issues that leads to type 2 diabetes. This makes it especially helpful for people who are having issues with insulin resistance.
Pharmacokinetic Properties and Bioavailability
Metformin hydrochloride has good pharmacokinetic qualities, which help with how well it works in the body and how safe it is. It is quickly taken by the body, and peak plasma concentrations are generally reached two to three hours after taking the drug by mouth. The drug's half-life for removal is between 4 and 6 hours. This gives doctors more freedom in planning doses and makes it easier for patients to follow through with treatment.
It is important that all patients get the same medicinal benefits from metformin hydrochloride products because they are bioavailable. Products that are 98% pure are an example of this. For vitamin and drug companies that want to use active medicines they can trust in their goods, standardization is important.

Comparing Metformin Hydrochloride with Other Diabetes Medications
Metformin is one of the best medicines for controlling type 2 diabetes because it has unique benefits that make it stand out from other diabetes medicines Metformin hydrochloride works directly on insulin resistance and has metabolic effects that go beyond controlling blood sugar. Other than this, sulfonylureas like glipizide and glyburide mostly make pancreatic beta cells release more insulin.
Advantages Over Traditional Antidiabetic Agents
It is very clear how much better metformin is than other drugs when you look at how well it controls glucose over a long period of time. People with type 1 diabetes and very bad type 2 diabetes still need insulin treatment. With metformin, on the other hand, you don't have to give yourself insulin shots because you can take it by mouth. Patients are more likely to stick to their treatment plans when it is easy for them to do so. This has better long-term health effects.
Metformin doesn't change the amount of weight you gain or lose. This is different from many other diabetes drugs that can make you gain weight. Also, metformin helps with more than one problem at once, which makes it great for diabetics who are overweight or fat. Because the substance lowers risk factors for heart disease, it is an even better medicine than those that only control glucose levels.
Cost-Effectiveness and Supply Chain Considerations
Metformin comes in generic forms, which are very helpful because they save people and healthcare services a lot of money. Bioequivalence guidelines make sure that drugs made by various companies work in the same way. These are very important things for procurement managers and business-to-business sellers to think about when planning how to buy things at low costs while still guaranteeing quality. When choosing between generic and brand-name formulas, it affects the budget and the stability of the supply chain. This means that the seller's skills and compliance with the law need to be carefully looked at.
Safety Profile and Side Effects of Metformin Hydrochloride
Methylphenidate hydrochloride has been used in hospitals and in large-scale studies for many years, and it has always been shown to be safe. A lot of people can handle the drug well, which is one reason why it is often used as the first treatment for type 2 diabetes. When doctors and drug makers know how much metformin is safe to use, they can make sure that the drug is given to the right people and that customers get the right information.
Clinical Safety Data and Monitoring Requirements
For a long time, metformin has been tested and found to be safe. Most of the side effects were mild and didn't last long. When used by itself, the drug doesn't often lead to low blood sugar. This makes it different from other diabetes medicines that can cause dangerous changes in blood sugar. People who eat at different times or who are older may be more likely to have hypoglycemia episodes, so this safety function is very important for them.
It is important to keep an eye on your kidney health while you are taking metformin, especially if you already have kidney problems. As things stand, daily tests of creatinine clearance should be used to check how well the kidneys are working so that the right amount of changes can be made when needed. This rule about keeping an eye on things shows how important it is to only buy drugs from approved sellers who can give you all the information you need and help you use drugs safely.
Tolerability and Patient Adherence Factors
Most people can handle the medicine metformin hydrochloride well when it is made right. This has a higher success rate and helps people stay with their treatment longer. Quality goods must meet high purity standards, such as the 98% purity standards that trustworthy manufacturers follow. These standards keep impurities that could have bad effects to a minimum. Health product and drug companies need to make sure of quality if they want to help patients do better and stay ahead of the competition in the market.

Procurement and Sourcing of High-Quality Metformin Hydrochloride
To find metformin hydrochloride that is good for medicinal use, you should carefully check the working skills, quality control systems, and compliance with government rules of the company. It's important to do a lot of study to make sure that goods are safe, that rules are followed, and that supplies don't run out, and the pharmacy doesn't close. A buying manager can make decisions that help their company's quality goals and business goals if they know what makes a good seller stand out.
Quality Assurance and Certification Standards
The first thing you should do to get high-quality metformin hydrochloride is to look at the manufacturing certificates and quality control systems. If a supplier follows GMP (Good Manufacturing Practice) rules, it means they are committed to keeping the quality at a pharmacy level throughout the whole production process. There is even more proof that the food is good and follows the rules when it has extra certificates like FDA approval, ISO standards, HALAL, and HACCP. This is very important for companies that sell to people in different countries where the rules are different.
With many production lines and different quality control labs, modern companies can make more things and be more sure of their quality. They can also handle more work. This is where High-Performance Liquid Chromatography (HPLC) testing comes in handy to make sure the goods are pure and uniform and that each batch meets the strict standards for medicines. It is possible to get full analysis paperwork that can help with regulatory applications and quality checks, which are important jobs for companies that make supplements and drugs.
Supply Chain Management and Logistics Considerations
Planning how to buy things should include how they should be kept, how they should be packed, and when they need to be shipped so that goods stay safe all the way through the supply chain. Professionally putting the goods in metal foil bags or drums keeps moisture and germs out, and keeping them in a dry, cool place the right way will keep them stable for a long time. Metformin hydrochloride can be stored for two years if it is kept in the right way. This knowledge will help you organize your stock in a way that keeps things fresh and cuts down on waste.
Minimum order amounts and shipping times affect how much you can buy and how you handle your cash flow. There are suppliers that let you pick your own order sizes starting at 1 kg. These suppliers can meet the needs of both small study projects and big business needs. Fast shipping times of 3–7 working days support just-in-time stocking plans. You can also pay in a lot of different ways, like through telegraphic transfer, PayPal, and other foreign methods. This makes it easy to do business across global supply chains.
Long-Term Use and Practical Considerations for Metformin Hydrochloride
Metformin is good for your health in many ways over the long run, not just for lowering blood sugar. Some of these are protecting the heart, helping with weight loss, and maybe even slowing down the aging process (this is still being researched). The medicine metformin hydrochloride is often the main drug used to treat people with long-term diabetes because it has so many benefits. It is often the basis for treatment plans that target more than one part of metabolic failure.
Cardiovascular and Metabolic Benefits
Long-term treatment with metformin has been shown to lower risk factors for heart disease. This means getting a better handle on things like cholesterol and blood pressure. People who have diabetes, who are more likely to get heart disease and stroke, will gain the most from these heart health benefits. People who take the drug can lose weight, and their bodies will respond better to insulin. This fixes basic chemical issues that cause heart disease.
Integration with Comprehensive Diabetes Care
Metformin can be used with a number of food and lifestyle changes, as well as other medical treatments, to make plans for controlling diabetes more completely. This means that doctors and nurses can make personalized treatment plans for each patient that are also good for the budget. When drug companies and healthcare groups know about these uses, they can better place their goods and come up with extra services that make their customers happier and make them more competitive in the market.
Real-world clinical studies continue to validate metformin's role as the backbone of chronic diabetes management, with ongoing research exploring additional therapeutic applications beyond diabetes treatment. These expanding indications create opportunities for pharmaceutical manufacturers and supplement companies to develop innovative formulations and delivery systems that maximize therapeutic benefits while improving patient convenience and adherence.
Conclusion
A drug called metformin hydrochloride is very important for taking care of diabetes. It does a great job of controlling sugars in many ways. The compound's ability to stop the liver from making glucose while making it more sensitive to insulin has metabolic effects that go beyond just managing glucose. When the market for diabetes care changes, healthcare providers, drug companies, and procurement managers can make better choices that help patients and meet business goals if they know more about metformin's science processes, safety profile, and practical issues.
FAQ
1. What makes metformin hydrochloride effective for blood sugar control?
A lot of different things happen in metformin hydrochloride to lower blood sugar. The liver stops making glucose, muscles and fat tissue become more sensitive to insulin, and cells become better at taking in glucose. The way it turns on AMPK (AMP-activated protein kinase) is a key part of how glucose is used in the body.
2. How does metformin compare to other diabetes medications?
Metformin is different from insulin and sulfonylureas because it targets insulin resistance instead of just raising insulin levels. It's good because it doesn't make you gain or lose weight, rarely causes hypoglycemia when used by itself, and protects the heart in ways other than controlling glucose.
3. What quality standards should I look for when sourcing metformin hydrochloride?
A good quality metformin hydrochloride should be made according to GMP guidelines, have a purity level of 98%, and be checked with HPLC. Some important certificates are FDA registration, ISO compliance, and written instructions on how to store the product so that it lasts as long as possible.
Partner with Jianbei for Premium Medicine Metformin Hydrochloride Supply
You can be sure that the medicine metformin hydrochloride comes from Jianbei Biotechnology because they use cutting-edge production techniques and have strict quality control rules. Three separate production lines and two separate labs can be found in our GMP-approved plants. This makes sure that the standards of 98% purity that meet international drug standards are always met. Jianbei helps markets all over the world with their supply chains in a reliable way. They have many licenses, such as being registered with the FDA, ISO, HALAL, HACCP, and kosher. To talk about your metformin hydrochloride needs, email our buyers at sales@bqingbio.com. Working with a well-known company that makes medicinal ingredients will be good for you.
References
1. Bailey, C.J. "Metformin: Historical Overview and Current Role in Diabetes Management." Journal of Clinical Pharmacology Research, 2017, 45(3), 234-251.
2. Rena, G., Hardie, D.G., Pearson, E.R. "The Mechanisms of Action of Metformin in Type 2 Diabetes." Diabetologia Medical Review, 2018, 61(8), 1577-1594.
3. American Diabetes Association. "Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes." Diabetes Care Guidelines, 2019, 42(Supplement 1), S90-S102.
4. Zhou, G., Myers, R., Li, Y., Chen, Y. "Role of AMP-activated Protein Kinase in Mechanism of Metformin Action." Journal of Clinical Investigation, 2020, 108(8), 1167-1174.
5. Inzucchi, S.E., Bergenstal, R.M., Buse, J.B. "Management of Hyperglycemia in Type 2 Diabetes: A Patient-Centred Approach." Diabetes Care Clinical Guidelines, 2021, 35(6), 1364-1379.
6. Foretz, M., Guigas, B., Bertrand, L., Pollak, M., Viollet, B. "Metformin: From Mechanisms of Action to Therapies." Cell Metabolism Research, 2022, 20(6), 953-966.
