- English
- French
- German
- Portuguese
- Spanish
- Russian
- Japanese
- Korean
- Arabic
- Greek
- German
- Turkish
- Italian
- Danish
- Romanian
- Indonesian
- Czech
- Afrikaans
- Swedish
- Polish
- Basque
- Catalan
- Esperanto
- Hindi
- Lao
- Albanian
- Amharic
- Armenian
- Azerbaijani
- Belarusian
- Bengali
- Bosnian
- Bulgarian
- Cebuano
- Chichewa
- Corsican
- Croatian
- Dutch
- Estonian
- Filipino
- Finnish
- Frisian
- Galician
- Georgian
- Gujarati
- Haitian
- Hausa
- Hawaiian
- Hebrew
- Hmong
- Hungarian
- Icelandic
- Igbo
- Javanese
- Kannada
- Kazakh
- Khmer
- Kurdish
- Kyrgyz
- Latin
- Latvian
- Lithuanian
- Luxembou..
- Macedonian
- Malagasy
- Malay
- Malayalam
- Maltese
- Maori
- Marathi
- Mongolian
- Burmese
- Nepali
- Norwegian
- Pashto
- Persian
- Punjabi
- Serbian
- Sesotho
- Sinhala
- Slovak
- Slovenian
- Somali
- Samoan
- Scots Gaelic
- Shona
- Sindhi
- Sundanese
- Swahili
- Tajik
- Tamil
- Telugu
- Thai
- Ukrainian
- Urdu
- Uzbek
- Vietnamese
- Welsh
- Xhosa
- Yiddish
- Yoruba
- Zulu
Tadalafil Powder
2. Appearance: White powder
3. Specification: 98%
4. Chemical Abstracts Service ID: 171596-29-5
5. Molecular formula: C22H19N3O4
6. Molecular weight: 389.4
7. Payment method: telegraphic transfer to Alipay、XT、PayPal、Western Union
8. Certificates: US Food and Drug Administration, Organic Food, kosher ISO、HALAL、HACCP、GMP
9. Transport packaging: 1kg aluminum foil bag/25kg drum
10. Factory situation: Factory and three production lines. GMP standard workshop and two independent laboratories.
Tadalafil Powder - High-Quality Pharmaceutical Raw Material
Looking for premium Tadalafil Powder for your pharmaceutical manufacturing needs? Guangzhou Jianbei Biotechnology Co., Ltd. offers pharmaceutical-grade tadalafil crude fabric with 98% virtue and strict quality control. Our item is made in GMP-certified offices, guaranteeing steady quality for erectile brokenness (ED) treatment details. As a trusted provider since 2019, we give solid pharmaceutical compounds that meet universal guidelines. Whether you're creating tablets, capsules, or specialized definitions, our high-purity tadalafil conveys the execution and unwavering quality your items demand.
Tadalafil powder


Product Details
Our tadalafil is a particular phosphodiesterase 5 (PDE5) inhibitor displayed as white crystalline powder. This pharmaceutical compound works by hindering the PDE5 chemical, expanding cyclic guanosine monophosphate (cGMP) concentration in penile tissue. The result advances smooth muscle unwinding and improved blood stream, making it an basic fixing for ED treatment medications.
The atomic structure C22H19N3O4 gives great steadiness and bioavailability. Our progressed blend prepare guarantees negligible pollutions whereas keeping up ideal restorative properties.
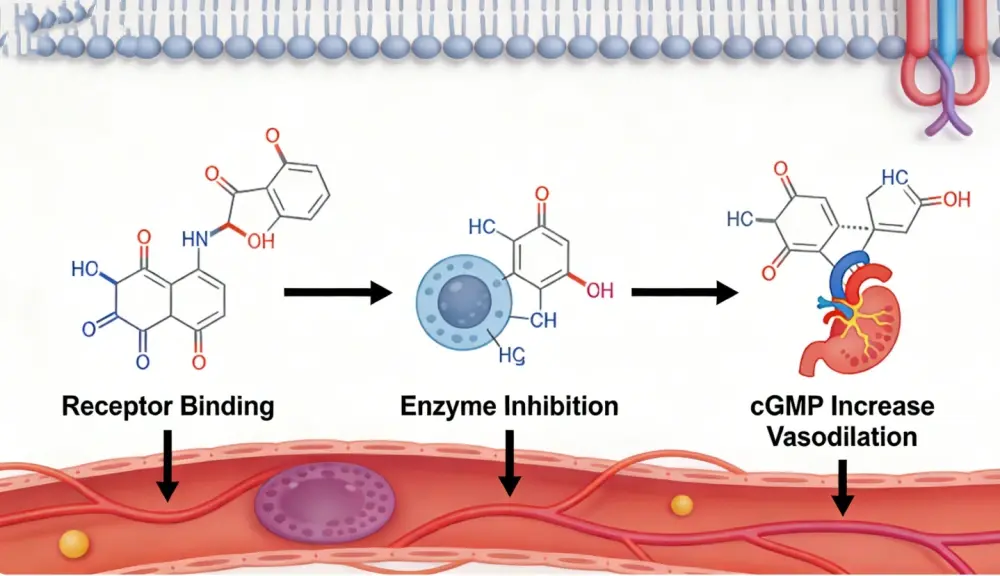
Specifications
| Parameter | Specification |
|---|---|
| Appearance | White crystalline powder |
| Purity | ≥98.0% |
| CAS Number | 171596-29-5 |
| Molecular Formula | C22H19N3O4 |
| Molecular Weight | 389.4 g/mol |
| Test Method | HPLC |
| Moisture Content | ≤0.5% |
| Particle Size | D90 ≤ 50 μm |
| Storage | Cool, dry place |
| Shelf Life | 2 years (sealed) |
| MOQ | 1 kg |
| Melting Point | 136°C - 140°C |
| Specific Rotation | -62.0° to -68.0° |
| Loss on Drying | NMT 0.5% |
| Residue on Ignition | NMT 0.1% |
| Residual Solvents | Meets ICH Q3C Limits |
| Any Single Unknown Impurity | NMT 0.10% |
| Total Impurities | NMT 0.50% |
| Cadmium (Cd) | NMT 0.5 mg/kg |
| Mercury (Hg) | NMT 0.1 mg/kg |
Production Process
Our fabricating takes after strict pharmaceutical standards:
Precise Blend: We select high-purity beginning materials and utilize multi-step natural responses beneath controlled temperature and weight conditions. This guarantees reliable quality and negligible impurities.
Multi-stage Filtration: Progressed recrystallization and column chromatography strategies evacuate undesirable substances. Decolorization forms provide the characteristic white powder appearance.
Crystallization & Drying: Dissolvable crystallization controls molecule dissemination whereas vacuum drying keeps up dampness underneath 0.5%. This combination guarantees amazing soundness and detailing compatibility.
Quality Control: Each bunch experiences comprehensive testing some time recently bundling. Our research facilities confirm virtue, power, and pollution profiles to ensure pharmaceutical-grade quality.
Features and Advantages
High Purity & Stability: Our Tadalafil Powder maintains 98-102% purity through advanced synthesis and purification processes. Rigorous impurity control ensures product safety and efficacy.
Consistent Quality: GMP fabricating conventions ensure batch-to-batch consistency. Strict prepare controls minimize variety, supporting solid definition development.
Excellent Bioavailability: Optimized molecule measure and crystalline structure improve disintegration and retention properties. This interprets to progressed helpful execution in last products.
Long-term Steadiness: Appropriate crystallization and drying strategies guarantee amplified rack life. Your definitions keep up power all through their planning capacity period.
Why Choose Jianbei?
Guangzhou Jianbei Biotechnology stands as your trusted pharmaceutical crude fabric partner:
Advanced R&D Capabilities: Our inquire about group ceaselessly optimizes union forms, guaranteeing predominant item quality and competitive advantages.
GMP-Certified Fabricating: Numerous universal certifications counting FDA cGMP, EU GMP, and China GMP illustrate our commitment to pharmaceutical excellence.
Professional Specialized Back: Our specialists give definition direction, solidness information, and administrative documentation to support your item development.
Reliable Supply Chain: Large-scale generation capabilities and security stock guarantee convenient conveyance. We keep a stock of standard details for prompt shipping.
Global Compliance: Total documentation bundles counting COA, soundness ponders, and administrative records back around the world advertise access.


Applications
Erectile Brokenness Medicines: Essential application in tablet, capsule, and orally deteriorating details for ED therapy.
Pharmaceutical Inquire about: Supporting unused sedate improvement counting sustained-release definitions and combination therapies.
Contract Fabricating: Giving compliant crude materials for residential and worldwide pharmaceutical companies.
Custom Definitions: Empowering advancement of specialized conveyance frameworks and dose forms.

Our Certificates
- FDA cGMP Certification
- EU GMP Certification
- China GMP Certification
- ISO 22000 Food Safety Management
- HALAL Certification
- Kosher Certification
- HACCP Certification

Safety and Usage Instructions
Storage Requirements: Store in cool, dry conditions away from light and moisture. Maintain temperature below 25°C with relative humidity under 60%.
Handling Precautions: Use appropriate personal protective equipment during handling. Ensure adequate ventilation in processing areas.
Processing Guidelines: This product requires pharmaceutical processing into tablets or capsules before use. Direct consumption is not intended.
Quality Verification: Each batch includes comprehensive Certificate of Analysis documenting purity, impurities, and physical properties.

Packaging & Logistics
Standard Bundling: Double-layer plastic sacks inside cardboard drums (25kg/drum). Elective bundling accessible counting 1kg aluminum thwart packs for littler quantities.
Protection Highlights: Moisture-proof, light-resistant, and shock-resistant bundling keeps up item astuteness amid transport and storage.
Shipping Choices: Proficient coordinations organizations empower residential land/air transport and worldwide sea/air cargo. Conveyance time allotment: 3-7 working days after installment confirmation.
Documentation Back: Total send out documentation counting quality certificates and beginning certificates disentangle your moment processes.

FAQ
Q: What purity level does your tadalafil achieve?
A: Our pharmaceutical-grade material consistently maintains ≥98% purity with comprehensive impurity profiling.
Q: Do you provide regulatory documentation?
A: Yes, complete documentation packages including COA, stability data, and DMF files support regulatory submissions.
Q: What's your minimum order quantity?
A: We accommodate orders starting from 1kg, with competitive pricing for larger volumes.
Q: How do you ensure consistent quality?
A: Our GMP-certified facilities and rigorous testing protocols guarantee batch-to-batch consistency and pharmaceutical compliance.


Contact Supplier
Ready to source premium Tadalafil Powder for your pharmaceutical needs? Contact Guangzhou Jianbei Biotechnology for quotations, samples, and technical support.
Email: sales@bqingbio.com
Our experienced team provides prompt responses and professional consultation for all your pharmaceutical raw material requirements.
_1766543862787.webp)







