- English
- French
- German
- Portuguese
- Spanish
- Russian
- Japanese
- Korean
- Arabic
- Greek
- German
- Turkish
- Italian
- Danish
- Romanian
- Indonesian
- Czech
- Afrikaans
- Swedish
- Polish
- Basque
- Catalan
- Esperanto
- Hindi
- Lao
- Albanian
- Amharic
- Armenian
- Azerbaijani
- Belarusian
- Bengali
- Bosnian
- Bulgarian
- Cebuano
- Chichewa
- Corsican
- Croatian
- Dutch
- Estonian
- Filipino
- Finnish
- Frisian
- Galician
- Georgian
- Gujarati
- Haitian
- Hausa
- Hawaiian
- Hebrew
- Hmong
- Hungarian
- Icelandic
- Igbo
- Javanese
- Kannada
- Kazakh
- Khmer
- Kurdish
- Kyrgyz
- Latin
- Latvian
- Lithuanian
- Luxembou..
- Macedonian
- Malagasy
- Malay
- Malayalam
- Maltese
- Maori
- Marathi
- Mongolian
- Burmese
- Nepali
- Norwegian
- Pashto
- Persian
- Punjabi
- Serbian
- Sesotho
- Sinhala
- Slovak
- Slovenian
- Somali
- Samoan
- Scots Gaelic
- Shona
- Sindhi
- Sundanese
- Swahili
- Tajik
- Tamil
- Telugu
- Thai
- Ukrainian
- Urdu
- Uzbek
- Vietnamese
- Welsh
- Xhosa
- Yiddish
- Yoruba
- Zulu
When working with Tadalafil Powder, the way you Tadalafil Powder do it relies on what you're doing, like making pharmaceutical products, doing research, or making specialised supplements. This very pure phosphodiesterase-5 (PDE5) inhibitor needs to be handled and measured very carefully. Manufacturers usually use controlled compounding methods to add tadalafil to tablet forms, capsules, or suspension systems. Analytical scales that have been calibrated must be used to correctly weigh the powder, and then it must be mixed with the right pharmaceutical ingredients to make sure that it is spread out evenly. Knowing the right way to reconstitute, figure out dosages, and think about stability helps keep this powerful active medicinal ingredient pure throughout the manufacturing process.
Understanding Tadalafil Powder: What Sets It Apart
Tadalafil is different from other PDE5 inhibitors because its half-life is longer, at about 17.5 hours. This pharmacokinetic profile means that the drug can work for up to 36 hours, which is a lot longer than other chemicals. The molecular structure (C22H19N3O4) helps it work for a long time, which makes it a popular choice for formulators who want to make long-lasting medicines. The white crystalline powder we work with at Jianbei meets strict pharmaceutical standards for clarity (98%). At this stage of refinement, impurities that could make the product less effective or cause safety concerns are removed. Our GMP-certified manufacturing processes guarantee consistency from batch to batch, which is very important when going from pilot runs to commercial volumes. This compound's lipophilic nature is what makes it so valuable; it affects both formulation strategies and bioavailability outcomes. Unlike chemicals that dissolve in water, tadalafil needs to be absorbed best in certain ways before it can be used.

Core Application Methods for Tadalafil Powder
Tablet Formulation Through Direct Compression
Adding tadalafil to solid tablet forms is easiest when it is done through direct compression. For this method, the active pharmaceutical ingredient is mixed with croscarmellose sodium to break it down and magnesium stearate to keep it from sticking. High-speed rotary tablet presses take the powder mixture and press it into tablets with the same amount of content. This is where the problem lies. Since tadalafil only makes up a small part of the tablet's weight (about 2.5 mg to 20 mg per unit), it is very important that the tablet is mixed well. Pharmaceutical companies use geometric dilution techniques, which involve gradually mixing smaller amounts of excipients with larger amounts, to keep the mixtures from separating during processing. This method solves the problem of production efficiency for big companies that need to make thousands of units per hour while still being able to do precise dosing. After that, film coatings can be put on the pills to make them more stable and help patients take them as prescribed.
Capsule Encapsulation for Custom Compounding
When working with tadalafil powder, compounding Tadalafil Powder shops often choose to make capsules. This method lets you change the dose easily, which is helpful for people who need non-standard strengths or who are allergic to certain ingredients in commercial tablet excipients. To do this, you have to figure out how much tadalafil you need and then mix it with harmless fillers like lactose monohydrate or microcrystalline cellulose to get the right capsule fill volume. A geometric diluting method makes sure that the powder blend is evenly spread out. The mixture is then put into empty vegetarian or gelatin capsules using either human or semi-automated capsule-filling equipment. This method solves the problem of personalisation in pharmaceutical care. Patients with benign prostatic hyperplasia may benefit from daily low-dose formulations (2.5 mg), while those who need medicine only when they need it may need stronger formulations. Compounders can make these different versions without having to buy various tablet production lines because capsules do it for them.
Suspension Systems for Enhanced Bioavailability
There are some special technical hurdles in making liquid suspensions from tadalafil powder because the compound doesn't like water. To make dispersions that stay solid, formulators need to add surfactants like polysorbate 80 or polyethylene glycol derivatives. The powder is first micronised to make the particles smaller. This increases the surface area and speeds up the dissolving process. Usually, micronised tadalafil is mixed with suspending agents like xanthan gum or carboxymethylcellulose to keep the mixture from settling. A process called high-shear mixing breaks up clumps in the mixture and makes a smooth solution. The formulation is finished with preservatives and flavourings that make it taste good and keep it fresh for a long time. This application method helps people who have trouble swallowing solid dosages, like kids (though not for normal tadalafil uses) or older people who have trouble swallowing. The liquid form also lets you precisely titrate the dose, which is helpful for clinical studies where researchers need the freedom to use exact doses.
Research-Grade Reference Standards
Labs that do analyses use tadalafil powder as a measure to help them create and confirm new testing methods. High-performance liquid chromatography (HPLC) methods need verified reference materials to set up retention times, calibration curves, and system suitability parameters. To make stock solutions, researchers weigh out exact amounts of the powder, usually in the milligram range, and dissolve it in the right solvents, such as methanol or acetonitrile. The standards are then diluted one at a time to make multi-point calibration curves that cover the concentration range that is expected in test samples. This application solves the quality control problem that generic makers and contract research organisations have. It is not possible to correctly measure the amount of active ingredients in finished products without using reference standards that have been checked. We keep the 98% purity standard at Jianbei to make sure that these standards give us accurate starting points for comparing things.
Sublingual Troche Production
Compounding specialists create sublingual troches by incorporating tadalafil powder into flavored base matrices that dissolve rapidly under the tongue. This delivery route bypasses first-pass hepatic metabolism, potentially improving bioavailability compared to oral tablets. The manufacturing process involves warming a polyethylene glycol base to its melting point, then dispersing the active ingredient throughout the molten mixture. Flavoring agents and sweeteners mask the compound's inherent bitterness. The mixture pours into troche molds and cools to room temperature, forming solid lozenges. This formulation strategy benefits patients who experience gastrointestinal side effects from conventional oral administration. The sublingual mucosa absorbs the active ingredient directly into systemic circulation, avoiding stomach acid exposure. Compounders appreciate this format for its ability to deliver therapeutic effects with reduced dosing requirements.
Transdermal Cream Compounding
Although less common, some compounding pharmacies Tadalafil Powder incorporate tadalafil into transdermal cream formulations. This approach requires specialized permeation enhancers like dimethyl sulfoxide (DMSO) or penetration-enhancing lipids to facilitate passage through the stratum corneum. The compounding process involves creating an oil-in-water emulsion base, then incorporating finely divided tadalafil powder along with permeation enhancers. Homogenization equipment ensures uniform particle distribution throughout the cream matrix. The finished product allows targeted application to specific anatomical areas. This delivery system addresses the discrete administration preference some patients express. Topical application eliminates the need for oral ingestion, which appeals to individuals taking multiple medications who wish to reduce pill burden. However, formulators must validate adequate transdermal absorption to ensure therapeutic levels reach systemic circulation.
Orodispersible Film Technology
Pharmaceutical innovators have developed thin films containing tadalafil that dissolve rapidly on the tongue. Production involves creating a polymer solution (typically using hydroxypropyl methylcellulose), dispersing micronized tadalafil throughout the solution, then casting the mixture into thin sheets using controlled-environment drying chambers. The dried films undergo precision cutting into individual dosage units, each containing an exact amount of active ingredient. Packaging in moisture-resistant pouches maintains stability throughout the product's shelf life. This modern application method solves both the portability and discretion challenges. The films require no water for administration, making them ideal for on-the-go use. The rapid dissolution provides a quick onset of action, which patients value when timing matters. Manufacturing efficiency remains high since automated casting and cutting equipment can produce thousands of units daily.
Essential Considerations When Handling Tadalafil Powder
Environmental controls play a crucial role in maintaining powder integrity. Our facility at Jianbei operates under controlled humidity conditions (below 60% relative humidity) because tadalafil exhibits hygroscopic tendencies. Moisture absorption can trigger degradation pathways that reduce potency over time. Temperature management matters equally. Storage in cool environments (below 25°C) preserves chemical stability. We maintain our inventory in climate-controlled warehouses where temperature fluctuations remain minimal. This attention to environmental factors ensures that the material you receive matches the specifications documented in our certificates of analysis. Light exposure presents another consideration. Tadalafil shows sensitivity to ultraviolet radiation, which can catalyze photodegradation. Opaque packaging materials—like amber glass containers or foil-lined bags—protect the powder during storage and transport. Our standard 1kg aluminum foil bags and 25kg drums provide this essential light barrier. Weighing accuracy cannot be overstated. Given the potent nature of this compound, even minor measurement errors translate to significant dosing discrepancies in finished products. Analytical balances with 0.1mg readability represent the minimum acceptable precision. Regular calibration using certified weights maintains measurement reliability across production batches. Cross-contamination prevention requires dedicated equipment for tadalafil processing. Manufacturers implementing multiple product lines should designate specific blenders, mills, and filling machines exclusively for PDE5 inhibitor formulations. This segregation prevents inadvertent transfer of active ingredients between incompatible product categories.
Quality Assurance and Testing Protocols
Validating the identity and purity of tadalafil powder begins the moment the material arrives at your facility. Incoming inspection should include visual examination for color uniformity and particle characteristics. The powder should appear consistently white without discoloration or clumping. Identity testing through Fourier-transform infrared spectroscopy (FTIR) confirms the molecular structure matches expected tadalafil spectra. This non-destructive technique compares the sample's infrared absorption pattern against authenticated reference standards. Spectral overlay with reference materials provides definitive identification. Quantitative analysis via HPLC determines actual purity levels. The method separates tadalafil from potential impurities using reversed-phase chromatography columns. UV detection at 283nm wavelength quantifies the compound based on peak area comparisons with calibrated standards. Results should consistently meet or exceed the 98% specification. Heavy metal screening ensures the powder meets pharmaceutical safety thresholds for lead, arsenic, mercury, and cadmium. Inductively coupled plasma mass spectrometry (ICP-MS) provides detection sensitivity in the parts-per-billion range, verifying compliance with ICH Q3D guidelines. Microbial testing confirms the absence of pathogenic organisms and verifies that total aerobic counts remain within acceptable limits. Standard plate count methods, yeast and mold enumeration, and specific pathogen testing (E. coli, Salmonella, Staphylococcus aureus) complete the microbial profile. At Jianbei, we conduct these comprehensive tests in our two independent laboratories before releasing any batch for distribution. Our quality management system, certified under ISO standards, documents every testing step to ensure traceability and regulatory compliance.
Regulatory Compliance Across Different Markets
Navigating the complex regulatory landscape requires understanding region-specific requirements. In markets across Southeast Asia, registration processes vary significantly between countries. Thailand's FDA equivalent requires detailed manufacturing documentation, while Indonesia emphasizes halal certification for products entering its pharmaceutical supply chain. Our HALAL certification addresses this requirement, opening doors to markets where religious compliance matters tremendously. This certification verifies that no prohibited substances contact the product during manufacturing and that production follows Islamic guidelines. Russian market entry demands registration with the Federal Service for Surveillance in Healthcare (Roszdravnadzor). The process requires comprehensive dossiers including manufacturing site inspections, stability data, and bioequivalence studies for generic Tadalafil Powder formulations. Our experience supplying materials to Russian pharmaceutical manufacturers has taught us the importance of maintaining complete documentation packages. South American markets, particularly Brazil and Argentina, have their own distinct requirements. ANVISA (Brazil's health authority) enforces strict GMP standards and requires extensive local testing before granting import authorization. Our FDA registration and multiple GMP certifications streamline this process, demonstrating our commitment to international quality standards. The European REACH regulation impacts anyone supplying materials into EU markets. This framework requires chemical safety assessments and registration of substances manufactured or imported above certain tonnage thresholds. Maintaining REACH compliance demonstrates environmental responsibility and facilitates market access across European nations.
Storage and Stability Management
When stored properly, Cialis powder can be used for a long time after it is first received. When handled properly, sealed containers kept in dry, cool places can stay stable for two years. At Jianbei, the aluminium foil packaging works well as a moisture barrier, but once it's opened, the food starts to be exposed to the world faster. Closing up opened containers right away after use keeps air from getting in. Some facilities use nitrogen purging, which fills the empty space inside containers with a harmless gas, to lower the oxidation potential even more. This method works especially well for bigger packages that are used up over long periods of time. In tropical climates like those found in Southeast Asia and parts of South America, changes in temperature during shipping can be dangerous. Thermal packaging options, like insulated shippers and phase-change cooling packs, keep the quality of the product safe during the 3–7 day shipping window. We plan shipments so that they don't get held up on the weekends at customs checkpoints, where the climate control could be lost. First-in, first-out (FIFO) inventory rotation keeps older items from sitting around in storage while younger ones are used up. This can be done easily because each package has a date code on it. This makes sure that the materials are used within their best stability window. Programs that track stability keep an eye on important quality traits over time. Usually, companies that make medicines do both short-term stability studies at normal conditions (25°C/60% RH) and long-term studies at higher temperatures (40°C/75% RH). These tools make data that supports dates for expiration and times for retesting.
Formulation Troubleshooting and Optimization
When formulators are making goods with tadalafil, they sometimes run into problems. The compound's hydrophobic nature often leads to problems with poor breakdown rates. To fix this, either the particle size needs to be smaller by micronising them or solubility-enhancing excipients like cyclodextrins need to be added. These make inclusion complexes that improve aqueous solubility. Problems with content uniformity during tablet production are often caused by not mixing the active ingredient and excipients well enough or by particle sizes not matching up correctly. Getting the tadalafil particles to the same median diameter as the filler materials helps them mix better and stops them from separating when the tablet is compressed. Stability failures, where the drug's effectiveness drops faster than expected, could mean that the active ingredient and the chosen excipients don't work well together. Some pill disintegrants, especially those that have a lot of water in them, can speed up the breakdown process. By doing compatibility studies during formulation development, it is possible to find mixtures that don't work well before starting full-scale production. Electrostatic interactions can sometimes cause coating defects on film-coated tablets. The chemical structure of tadalafil can create static charges during processing, which can cause the coating to be spread out unevenly. These problems can be fixed by controlling the humidity in the environment and adding antistatic agents to the coating suspension. At Jianbei, our expert team helps customers with these formulation problems. We offer advice that goes beyond just providing raw materials because we have 15 national invention patents that cover key processes and years of experience in manufacturing.
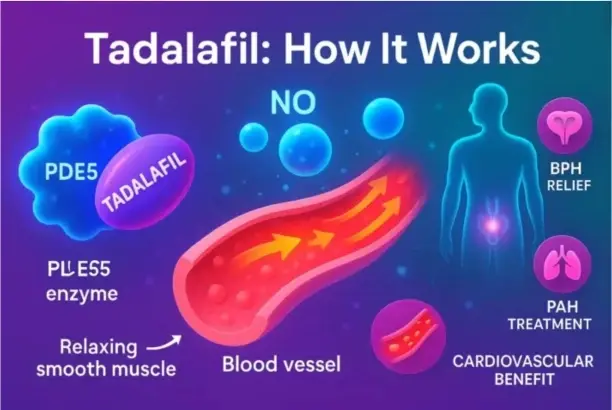
Conclusion
To work with Cialis powder, you need to know about technology, quality, and regulations. Each application method, from pill compression to suspension formulation, is made to fit the needs of the manufacturer and the patient. Handling guidelines, environmental controls, and thorough testing make sure that finished goods always have the same therapeutic effects. The pharmaceutical industry is always changing with new delivery methods that make it easier for patients to follow their treatments and make them work better. Formulators can safely make products that meet market needs and regulatory requirements in a wide range of international areas if they understand these basic ideas and work with qualified suppliers who uphold strict quality standards.
FAQ
1. What equipment do I need for weighing Tadalafil Powder accurately?
An analytical balance with at least 0.1mg readability is essential for precise measurement. The balance should sit on a vibration-dampening table in a draft-free environment. Regular calibration using certified reference weights maintains accuracy. Many pharmaceutical operations use balances with 0.01mg readability when formulating low-dose products.
2. Can Tadalafil Powder be mixed directly with water?
The compound exhibits poor water solubility due to its lipophilic structure. Direct mixing with water produces suspensions rather than true solutions. Achieving stable liquid formulations requires surfactants like polysorbate 80 or cosolvents such as polyethylene glycol. Micronization of the powder before dispersion improves suspension stability.
3. How do I calculate the correct amount for a specific dosage?
Dosage calculations require knowing the target strength per unit and the total number of units. For example, producing 1000 tablets at 10mg strength requires 10,000mg (10g) of tadalafil powder at 100% purity. When working with 98% purity material, divide the target amount by 0.98 to determine the actual quantity needed (10.2g in this example). Always account for overage to compensate for processing losses.
Partner with a Trusted Tadalafil Powder Manufacturer
Jianbei Biotechnology combines advanced R&D capabilities with reliable GMP manufacturing to serve health supplement companies worldwide. Our large inventory means your Tadalafil Powder ships within 3-7 working days, avoiding production delays that impact your market timelines. As an established supplier with complete FDA, ISO, HALAL, and HACCP certifications, we understand the compliance challenges you face in Southeast Asian, Russian, and South American markets. Connect with our technical team at sales@bqingbio.com to discuss your specific formulation requirements and discover how our 98% purity pharmaceutical-grade material supports your product development goals.
References
1. Pharmaceutical Manufacturing Encyclopedia: Active Pharmaceutical Ingredients in Solid Dosage Forms, Third Edition, William Andrew Publishing, 2015
2. Handbook of Pharmaceutical Excipients, Eighth Edition, Pharmaceutical Press and American Pharmacists Association, 2017
3. Remington: The Science and Practice of Pharmacy, Twenty-Second Edition, Pharmaceutical Press, 2013
4. Pharmaceutical Dosage Forms: Tablets, Volume 1: Unit Operations and Mechanical Properties, Third Edition, Informa Healthcare, 2008
5. Quality Assurance in the Pharmaceutical Industry: Best Practices for GMP Compliance, Elsevier Science Publishing, 2016
6. International Conference on Harmonisation Q3D Guideline: Elemental Impurities, ICH Expert Working Group, 2014
