- English
- French
- German
- Portuguese
- Spanish
- Russian
- Japanese
- Korean
- Arabic
- Greek
- German
- Turkish
- Italian
- Danish
- Romanian
- Indonesian
- Czech
- Afrikaans
- Swedish
- Polish
- Basque
- Catalan
- Esperanto
- Hindi
- Lao
- Albanian
- Amharic
- Armenian
- Azerbaijani
- Belarusian
- Bengali
- Bosnian
- Bulgarian
- Cebuano
- Chichewa
- Corsican
- Croatian
- Dutch
- Estonian
- Filipino
- Finnish
- Frisian
- Galician
- Georgian
- Gujarati
- Haitian
- Hausa
- Hawaiian
- Hebrew
- Hmong
- Hungarian
- Icelandic
- Igbo
- Javanese
- Kannada
- Kazakh
- Khmer
- Kurdish
- Kyrgyz
- Latin
- Latvian
- Lithuanian
- Luxembou..
- Macedonian
- Malagasy
- Malay
- Malayalam
- Maltese
- Maori
- Marathi
- Mongolian
- Burmese
- Nepali
- Norwegian
- Pashto
- Persian
- Punjabi
- Serbian
- Sesotho
- Sinhala
- Slovak
- Slovenian
- Somali
- Samoan
- Scots Gaelic
- Shona
- Sindhi
- Sundanese
- Swahili
- Tajik
- Tamil
- Telugu
- Thai
- Ukrainian
- Urdu
- Uzbek
- Vietnamese
- Welsh
- Xhosa
- Yiddish
- Yoruba
- Zulu
With the release of semaglutide, a peptide-based drug known around the world for its ability to manage type 2 diabetes and obesity, the pharmaceutical industry has seen huge progress. Understanding what the raw material for semaglutide is is important for buying professionals, R&D leaders, and product managers in the pharmaceutical and nutraceutical businesses who have to find active pharmaceutical ingredients. Semaglutide raw powder is the main ingredient that is used to make injectable medicines sold under different brand names. It is of a medicinal grade. This detailed guide talks about the chemical makeup, quality standards, buying factors, and production rules that are very important for B2B buyers who want to find trustworthy, legal, and pure peptide ingredients. Learning about this important raw material can help you make smart choices about where to get it and build partnerships based on quality and compliance, whether you're making new health supplements, useful foods, or pharmaceutical formulations.
Understanding Semaglutide Raw Powder
Semaglutide raw powder is the pharmaceutical-grade peptide material that has been lyophilized. It is the active pharmaceutical ingredient (API) in drugs that help control blood sugar and weight. This biochemical is a synthesized peptide with the CAS number 910463-68-2. It is a glucagon-like peptide-1 (GLP-1) receptor agonist. Its molecular formula, C187H291N45O59, shows that it has a complicated structure and a molecular weight of 4113.58 g/mol. This man-made peptide works a lot like the incretin hormones that are naturally made in the gut. These hormones are very important for controlling insulin production and hunger.
An aminoisobutyric acid replacement at position 8 and a fatty acid side chain addition at position 26 change the structure of semaglutide. These changes were made on purpose to fix problems with the stability of native GLP-1, which is quickly broken down by dipeptidyl peptidase-4 (DPP-4). A large increase in the peptide's half-life, to about 165 hours, is caused by acylation with a C-18 fatty diacid chain. This allows strong, reversible binding to serum albumin. This longer period lets dosing happen once a week, which greatly improves patient obedience and therapeutic results.
Mechanism of Action and Therapeutic Value
Semaglutide works by attaching to GLP-1 receptors found in beta cells in the pancreas, the digestive tract, and certain parts of the brain that control hunger. When activated, these receptors increase messages that tell the body it's full, stop the release of unnecessary glucagon, slow down stomach emptying, and increase insulin secretion that depends on glucose. This leads to a better metabolism in many ways, including stable blood sugar levels with fewer post-meal spikes, long-term weight loss by reducing hunger, and effects that protect the heart. Because of these health benefits, semaglutide is a key ingredient in products that aim to improve metabolic health. This makes the raw powder very useful for companies that make medicines and supplements for managing weight and taking care of diabetics.
Production Methodology
Recombinant DNA technology or solid-phase peptide synthesis (SPPS) is used to make high-quality semaglutide base material. These cutting-edge biotechnological methods make sure that the exact sequence of amino acids and post-translational changes are made that are needed for biological function. Manufacturers usually make the peptide in lyophilized form, which is a white to off-white hygroscopic powder. This makes it more stable on the shelf and easier to work with while formulating. The lyophilization method gets rid of the water content of the peptide while keeping its structure intact. This lets it be stored for longer periods of time under controlled conditions.
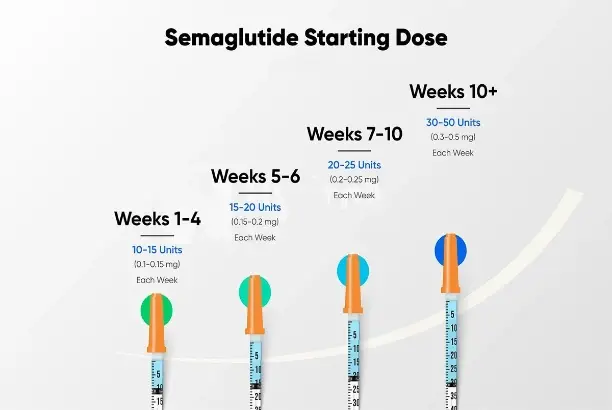
Key Characteristics and Quality Parameters of Semaglutide Raw Powder
When buying peptide-based active ingredients, including Semaglutide raw powder, quality control is still the most important thing. The purity and security of the raw material have a direct effect on how well, safely, and legally the final product works. Understanding important quality factors helps procurement managers rate suppliers well and make sure that performance is uniform from batch to batch.
Chemical Composition and Purity Standards
High-performance liquid chromatography (HPLC), the usual analytical method for peptide analysis, shows that pharmaceutical-grade semaglutide raw powder is at least 98% pure. Premium sellers usually send products that are more than 99% pure, with flaws kept to less than 0.5%. D-amino acid isomers, deletion peptides, and other similar substances that come from synthesis or breakdown processes are important pollutants that need extra care. Nitrogen research shows that the peptide content usually meets or exceeds 85%, which means that the amino acids are properly incorporated.
The white, powdery look means that the lyophilization went well and there was no reactive breakdown. Any color change indicates bad handling or storage conditions. The moisture level should stay below 5% to keep things from sticking together and to keep them stable over time. The International Council for Harmonisation (ICH) says that residual solvents from synthesis processes must follow its rules. This makes sure that the end product is safe.
Stability and Storage Considerations
Proper keeping conditions have a direct effect on how long peptide raw materials last and how well they work. When kept at -20°C in airtight, moisture-resistant cases out of the way of the sun, semaglutide raw powder maintains its ideal stability. Materials stay fully effective for up to 24 months when kept in these settings. Keeping things in dry, cool places stops them from breaking down in water and clumping together, which are both bad for biological activity.
Handling procedures should keep people from being exposed to changes in temperature and humidity as little as possible. Suppliers who pack their goods in 1kg aluminum foil bags or 25kg fiber drums with nitrogen cleaning give them extra protection against oxidative stress and water damage. These packaging techniques meet the needs of the pharmaceutical cold chain and make foreign shipping safer.
Regulatory Compliance and Documentation
Global business-to-business purchases need a lot of legal paperwork to show that the products are of good quality and that the production process follows the rules. GMP (Good Manufacturing Practice), ISO standards, HACCP (Hazard Analysis and Critical Control Points), HALAL, and kosher licenses are some of the most important ones. Materials that are going to be sold in the United States can be safer if they are registered with the FDA.
A Certificate of Analysis (COA) should be sent with each batch that lists important quality factors, such as the percentage of purity, the amount of peptides, the amount of heavy metals, the number of microbes that can't grow, and the level of endotoxin. Material Safety Data Sheets (MSDS) list how to handle, what to do in an emergency, and how to get rid of a substance safely. These papers back up government filings and show that the provider is dedicated to quality and openness.
Procurement Guide for Semaglutide Raw Powder
To find pharmaceutical-grade peptide materials, you need to carefully evaluate suppliers, communicate your needs clearly, and plan your processes well. This framework for purchasing deals with problems that many foreign buyers have when they try to find their way through complicated supply lines.
Supplier Selection Criteria
Checking the manufacturing skills and quality systems of potential providers is the first step in finding reliable ones. GMP-certified facilities show they follow the rules for making medicines, like keeping the surroundings safe, making sure the process works, and keeping lots of records. Suppliers that offer specialized peptide synthesis and have their own analysis labs make sure that quality is always the same and that batches are tested quickly.
Through its integrated method, Jianbei Biotechnology is a great example of a seller. The company uses advanced biotechnology equipment and two separate quality control laboratories to run three production lines in a GMP-standard workshop in Guangzhou Software Park. This setup allows for strict testing procedures and quick turnaround times. The company has 15 national discovery patents and 8 utility model patents, which show that it has the technical know-how and ability to come up with new ideas for peptide production and purification.
Minimum Order Quantities and Pricing
When buying industrial peptides, you usually have to meet a minimum order quantity (MOQ) that is based on the costs of synthesis batches and the standards for packing. For Semaglutide raw powder, the minimum order quantity (MOQ) is usually 1 kg, which is enough for both small-scale recipe research and mass production. This easy-to-reach level lets both new nutraceutical businesses and well-known drug companies get the materials they need for the amount of output they need.
Through tiered discounts, bulk pricing systems reward customers who make bigger purchases. Buyers should ask for thorough quotes that include price breaks at different volume levels, payment terms, and any extra costs that may apply for fast production or special tests. Clear conversations about prices build trust and make it possible to accurately model costs for budgets for product development.
Payment Methods and Terms
For international deals to go smoothly, payment systems need to be able to adapt to different financial systems and risk choices. Reliable sellers accept a number of payment methods, such as Western Union, PayPal, Alipay, and telegraphic transfer. This makes it easier for buyers in different parts of the world to buy from them. During the contract discussion process, clear payment terms should be set. These could include prepayment, letters of credit, or staged payments tied to production goals.
Commercial invoices, packing lists, certificates of origin, and customs statements are all types of paperwork that help with foreign trade. Suppliers who have experience with exporting offer complete paperwork packages that make it easier to clear customs and follow rules in the target country.
Logistics and Delivery Considerations
To keep the cold chain intact and keep the materials from being exposed to bad weather, peptide materials need to be handled in a certain way when they are shipped internationally. Temperature-sensitive items are kept safe during transport in dry ice packaging or cold cases. For established suppliers with efficient logistics partnerships, delivery times are usually between 3 and 7 working days. However, foreign shipping may take longer based on where the package is going and how long it takes to clear customs.
Buyers should clear up who is responsible for shipping, what insurance covers, and who is responsible for what. When you buy something, the words FOB (Free on Board), CIF (Cost, Insurance, and Freight), or DDP (Delivered Duty Paid) tell you who is responsible for paying for the shipping costs, insurance, and customs taxes. A clear understanding of these terms keeps disagreements and extra costs from happening.
Partner with Jianbei: Your Trusted Semaglutide Raw Powder Supplier
Choosing the right peptide provider has a direct effect on the success of the product, compliance with regulations, and the ability to compete in the market. Jianbei Biotechnology stands out because it has integrated skills that include research, manufacturing, and quality assurance. This makes us a preferred partner for global B2B procurement workers who need reliable access to high-quality semaglutide raw powder.
Our commitment to greatness in manufacturing is based on strict quality control methods that are backed up by a wide range of certifications. Along with our FDA registration, we also have ISO, HALAL, HACCP, and GMP licenses, which show that we follow international standards for pharmaceuticals. Every production batch goes through a full HPLC study that confirms at least 98% purity, and most batches are purer than 99%. Each package comes with detailed COA paperwork that makes important quality factors like peptide concentration, impurity profiles, and microbial limits clear.
In addition to production, we also offer unique solutions that meet the needs of a wide range of clients. Through custom formulations, package layouts, and legal support, our OEM and private label services help brands stand out. Our expert team works closely with customers from the initial idea to the final product, whether they are making unique supplement lines, pharmaceutical preparations, or functional food ingredients. Order amounts that start at 1 kg are flexible enough to meet the needs of both product creation and large-scale production.
Logistics efficiency makes sure that goods get to customers quickly and in great shape. Our efficient production schedule and established shipping partnerships allow us to offer standard arrival times of 3–7 business days. Shipments are made in protective 1kg aluminum foil bags or 25kg drums with moisture barriers and temperature tracking. This keeps the purity of the products safe while they are traveling across foreign borders. Customers all over the world can easily make purchases because there are many ways to pay, such as telegraphic transfer, PayPal, and Alipay.
Our dedication goes beyond delivering products, including Semaglutide raw powder; it also includes full procurement help. Account managers are responsible for keeping you informed about the progress of your orders, giving you expert advice, and helping you after the sale. Requests for samples let formulas be tested before promises to buy in bulk, which lowers the risk of buying. This focus on the client leads to long-lasting relationships based on trust, consistent quality, and shared success.

Conclusion
Procurement professionals can make smart buying choices that are in line with quality goals, legal requirements, and business goals when they know what the basic materials for semaglutide are. Semaglutide raw powder is an advanced GLP-1 receptor agonist peptide that is made through exact molecular engineering. It is the main ingredient in many pharmaceutical and nutritional products that help people with diabetes and weight loss. Critical quality factors, such as a minimum purity of 98%, the right way to store materials, and full legal paperwork, make sure that materials meet pharmaceutical-grade standards. When makers look at the strategic benefits of buying raw powder versus finished goods and other peptides, they can see how they can control the formulation and cut costs. To choose a good provider, you should put GMP certification, analytical skills, transportation knowledge, and clear communication at the top of your list. Companies that are ahead of the curve will stay ahead of the competition as the metabolic health market grows by making sure they can always get high-quality peptide materials.
FAQ
1. What purity level should I expect from pharmaceutical-grade semaglutide raw powder?
According to HPLC, pharmaceutical-grade material should be at least 98% pure, and the best providers always give 99% purity or higher. Individual impurities must be kept below 0.5%, especially important ones like deletion sequences and D-amino acid isomers. The nitrogen study should show that the peptide content is higher than 85%, which means that the amino acids are properly incorporated.
2. How should semaglutide raw powder be stored to maintain potency?
For best keeping, keep sealed packages at -20°C in dry, cool places that aren't exposed to light. When kept in these settings, things stay effective for 24 months. Putting things in aluminum foil bags and filling them with nitrogen gives them extra protection against moisture and rust. Long-term stability is maintained by limiting exposure to changes in temperature and humidity during handling.
3. Can semaglutide raw powder be used for both injectable and oral formulations?
Raw powder gives formulators more options for making medicines that work with different transportation methods. New research is looking into oral versions that use absorption boosters and protective coatings. Current market goods focus on injectable treatment. Getting raw materials lets companies come up with new ways to give medicines that meet the changing needs of patients and the tastes of the market.
Contact Jianbei Biotechnology for Premium Semaglutide Raw Powder
Jianbei Biotechnology is a good choice for pharmaceutical and nutraceutical businesses that need a reliable semaglutide raw powder producer. Our GMP-certified facilities, wide range of testing options, and experienced research and development team make sure that you can always get high-purity peptide ingredients that meet international standards. Our large inventory allows us to complete orders quickly, and shipping times of 3–7 business days support flexible production scheduling. We are committed to providing you with the best technical support and quality goods, whether you are making revolutionary weight loss pills, diabetes care products, or heart health solutions. Email our purchasing team at sales@bqingbio.com to talk about your particular needs, ask for samples of our products, or look into custom OEM partnerships. Find out how Jianbei's track record, patent-protected processes, and customer-focused service model make us your best choice for long-term supply of important medicinal raw materials.
References
1. Lau J, Bloch P, Schäffer L, et al. Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide. Journal of Medicinal Chemistry, 2015.
2. Nauck MA, Petrie JR, Sesti G, et al. A Phase 2, Randomized, Dose-Finding Study of the Novel Once-Weekly Human GLP-1 Analog, Semaglutide, Compared With Placebo and Open-Label Liraglutide in Patients With Type 2 Diabetes. Diabetes Care, 2016.
3. Wilding JPH, Batterham RL, Calanna S, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. New England Journal of Medicine, 2021.
4. Marso SP, Bain SC, Consoli A, et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. New England Journal of Medicine, 2016.
5. Kalra S, Baruah MP, Sahay RK, et al. Semaglutide: A Comprehensive Clinical Review. Diabetes Therapy, 2021.
6. Dhillon S. Semaglutide: A Review in Type 2 Diabetes. Drugs, 2018.
