- English
- French
- German
- Portuguese
- Spanish
- Russian
- Japanese
- Korean
- Arabic
- Greek
- German
- Turkish
- Italian
- Danish
- Romanian
- Indonesian
- Czech
- Afrikaans
- Swedish
- Polish
- Basque
- Catalan
- Esperanto
- Hindi
- Lao
- Albanian
- Amharic
- Armenian
- Azerbaijani
- Belarusian
- Bengali
- Bosnian
- Bulgarian
- Cebuano
- Chichewa
- Corsican
- Croatian
- Dutch
- Estonian
- Filipino
- Finnish
- Frisian
- Galician
- Georgian
- Gujarati
- Haitian
- Hausa
- Hawaiian
- Hebrew
- Hmong
- Hungarian
- Icelandic
- Igbo
- Javanese
- Kannada
- Kazakh
- Khmer
- Kurdish
- Kyrgyz
- Latin
- Latvian
- Lithuanian
- Luxembou..
- Macedonian
- Malagasy
- Malay
- Malayalam
- Maltese
- Maori
- Marathi
- Mongolian
- Burmese
- Nepali
- Norwegian
- Pashto
- Persian
- Punjabi
- Serbian
- Sesotho
- Sinhala
- Slovak
- Slovenian
- Somali
- Samoan
- Scots Gaelic
- Shona
- Sindhi
- Sundanese
- Swahili
- Tajik
- Tamil
- Telugu
- Thai
- Ukrainian
- Urdu
- Uzbek
- Vietnamese
- Welsh
- Xhosa
- Yiddish
- Yoruba
- Zulu
Semaglutide raw powder represents a high-purity active pharmaceutical ingredient (API) that has revolutionized the landscape of metabolic health formulations. This white crystalline compound, with the chemical formula C187H291N45O59 and molecular weight of 4113.58, serves as the foundational element for manufacturing diabetes management and weight control supplements. Understanding this sophisticated glucagon-like peptide-1 (GLP-1) receptor agonist becomes essential for B2B procurement professionals seeking reliable, standardized ingredients that meet stringent international quality standards and regulatory compliance requirements.
Understanding Semaglutide Raw Powder
Chemical Properties and Structure
Through its complicated molecular structure, the active ingredient shows a high level of biochemical complexity. With the Chemical Abstracts Service ID 910463-68-2, this substance is very stable when kept in the right way, in a dry, cool place. The semaglutide raw powder stays effective thanks to a carefully made peptide chain that works like natural incretin hormones. This makes insulin work better with glucose and improves the body's ability to control blood sugar.
High-Performance Liquid Chromatography (HPLC) methods are used in modern scientific testing to make sure that purity standards are met at concentration levels of 98%. This analytical accuracy makes sure that there is consistency from batch to batch, which is very important for pharmaceutical-grade uses. This helps makers keep their products working and meet the documentation needs of regulators in many different foreign markets.
Mechanism of Action
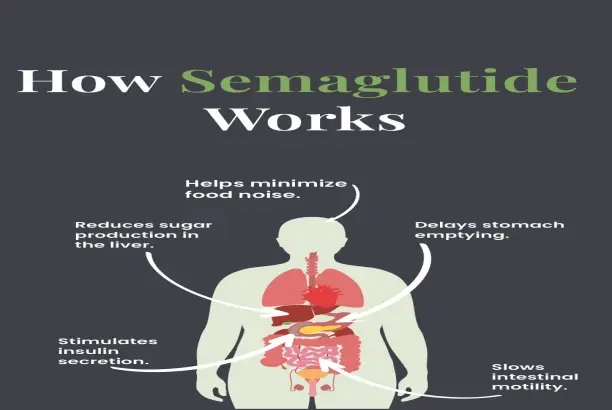
Agonism of the GLP-1 receptor is the main therapeutic route by which this drug improves metabolism. The ingredient turns on beta cells in the pancreas, which increases insulin production, while also stopping alpha cells from releasing glucagon. This two-part system provides a balanced approach to glucose control that works especially well for dealing with problems related to post-meal hyperglycemia.
Beyond its effects on blood sugar, the substance changes how quickly the stomach empties and encourages signals of fullness in the brain and spinal cord. These processes help people lose weight in a big way, which makes the ingredient useful for making complete metabolic health products that hit multiple therapeutic targets in a single formulation method.
Applications in Product Development
Manufacturing uses include making useful powders for immune-boosting, antioxidant, and weight control formulas, among other types of supplements. The ingredient works well in a number of different delivery methods, from standard capsules to new powder blends that stay stable during storage and processing.
Protocols for quality assurance make sure that FDA registration requirements, REACH standards, and ISO approval steps are followed. Following these thorough safety steps gives makers the peace of mind to add the ingredient to their products, keeping their competitive edge in regulated markets across North America and other countries.
Comparative Analysis of Semaglutide Raw Powder
Efficacy Benchmarks
A clinical study shows that these ingredients work better than standard metabolic support ingredients. Studies show big gains in lowering HbA1c, with subjects' glucose levels staying stable for long amounts of time during treatment. The semaglutide raw powder has reactions that change with dose, which allows formulators to change the level of potency based on the needs of the target population and the treatment goals.
Studies on the effectiveness of weight management show that people lose a lot of body fat and keep it off for a long time if they make the right changes to their lifestyle. These results support strategies for differentiating products for supplement makers who want to make marketing claims that are backed up by strong scientific proof and peer-reviewed research papers.
Purity and Quality Standards
Higher purity products are directly linked to better treatment results and lower levels of adverse reactions. The 98% standard makes sure that there aren't many impurities and that the active compounds are bioavailable to the body as much as possible. Heavy metals, microbial toxins, and leftover liquids that could affect the safety or effectiveness of a product are removed by advanced purification methods.
Manufacturing sites that follow GMP guidelines follow strict quality control rules during all stages of production. Testing in a separate lab makes sure that each batch meets the standards for consistency, stability, and lack of contamination. This gives procurement managers all the information they need to support regulatory reports and foreign export requirements.
Cost-Performance Analysis
When you compare treatment potency levels to cost-per-gram measures, the benefits of buying in bulk become clear. The high activity concentration of the ingredient lets makers get the desired formulation strengths with smaller amounts than with other compounds. This lowers total production costs while keeping the premium product positioning.
Transparency in the supply chain makes it possible to accurately predict prices and plan budgets for medium- to large-scale production runs. Minimum order quantities of 1 kg are set to suit both pilot-scale development projects and commercial manufacturing needs. This makes it easier to use flexible purchasing strategies that fit the needs of different businesses and the timetables for market growth.
Procurement Guide for Semaglutide Raw Powder
Sourcing Considerations
Reliable sellers keep a full set of certifications, such as FDA registration, organic certifications, kosher and HALAL compliance, as well as HACCP and GMP production standards. These certificates make sure that different foreign markets follow the rules, and they also back up product claims and marketing positioning strategies for networks that sell goods all over the world.
Specifications for packaging use metal foil bags for 1 kg orders and drum cases for 25 kg orders. This keeps the product's integrity while it's being shipped and stored. Protective packaging stops moisture absorption, light exposure, and contamination risks that could make ingredients less stable or effective over long periods of time.
Logistics and Storage Requirements
For proper keeping, items must be kept in dry, cool places that maintain the security of the ingredients for the two years that they are sealed. Temperature control systems and humidity tracking tools keep things from breaking down and keep the right conditions for long-term inventory management and quality preservation practices.
Delivery times are usually between 3 and 7 working days after payment is confirmed. This makes it easy to plan production and keep track of supplies. There are many ways to pay, such as telegraphic transfer, Alipay, PayPal, and Western Union. These payment choices make transactions more flexible and meet the needs of foreign buyers and financial processors.
Quality Verification Protocols
Before making big purchases, HPLC testing methods provide clear purity proof and impurity identification. Manufacturers can use sample testing tools to make sure that ingredients work with current formulation systems and that performance traits are valid in real production settings.
There are certificates of analysis, production records, and regulatory compliance checks for Semaglutide raw powder in documentation packages that help with audit standards and the regulatory submission process. Throughout the supply chain management process, full tracking systems keep track of where ingredients come from, how they are processed, including Semaglutide raw powder, and where quality control checks are done.
Selecting the Right Semaglutide Raw Powder Supplier
Certification and Compliance Standards
Leading suppliers show their dedication to quality by achieving multiple certifications that cover a wide range of foreign legal regimes. FDA registration makes sure that the product meets the standards of the US market, and ISO licenses show that the production process is consistent and the quality management system works well across all activities around the world.
HACCP and GMP licenses give you more peace of mind about how to keep your products safe, avoid contamination, and keep your processes under control. These standards are especially important for companies that want to make pharmaceutical-grade products or get permission to make therapeutic claims in controlled supplement categories and functional food uses.
Manufacturing Capabilities Assessment
To figure out a factory's production potential, you have to look at its infrastructure, such as the number of production lines, lab facilities, and quality control tools. Advanced makers run several production lines in GMP-compliant workplaces that are backed up by separate labs that can do a wide range of tests and ensure the quality of the products.
As part of evaluating technical knowledge, you should look at things like research and development capabilities, formulation support services, and custom manufacturing choices. Suppliers with strong research and development (R&D) teams offer helpful technical advice during all stages of product development, helping with efforts to improve performance and fixing problems that come up during the creation process.
Partnership Reliability Factors
For long-term supply agreements to work, sellers must have a track record of managing inventory well and delivering on time. Semaglutide raw powder providers should keep enough stock on hand to keep production running smoothly and offer flexible order scheduling to account for changes in yearly demand and the need for the market to grow.
When dealing with complicated rules, technical details, and foreign shipping methods, the quality of customer service becomes very important. In competitive buying settings, responsive communication, technical knowledge, and the ability to solve problems set trusted partners apart from transactional providers.
Clinical and Market Insights on Semaglutide Raw Powder
Evidence-Based Efficacy Data
More than just managing glucose, recent clinical studies show that semaglutide raw powder offers important cardiovascular protection effects. Research shows lower chances of major adverse cardiovascular events. This supports wider therapeutic claims and more market placement possibilities for supplement makers who want to offer complete metabolic health solutions.
Weight loss research data shows that over 68 weeks of treatment, people lost an average of 10-15% of their starting body weight. These results back up marketing claims based on evidence and give supplement companies a scientific basis for positioning expensive products and coming up with beneficial ways to stand out in the supplement market.
Market Demand Trends
As people learn more about digestive health, the demand for ingredients backed by science and clinically proven to work goes up. The market for semaglutide raw powder is growing because more people are becoming overweight, people are living longer, and healthcare is focusing more on preventive methods instead of reactive treatment methods.
As regulations change, they keep adding more allowed uses and therapeutic recommendations. This opens up more market possibilities for new products that target specific groups of people. These trends help producers plan their long-term purchases and investments in inventory so they can take advantage of new market possibilities.
Future Applications and Innovations
As the research stream grows, it looks into new delivery methods, combination treatments, and therapeutic uses that go beyond standard metabolic health categories. These new developments give makers who are willing to think ahead the chance to make next-generation formulas that are in line with how the market will change and what consumers want.
New developments in formulation technology have led to better bioavailability, less frequent doses, and better patient cooperation. These changes help elite product placement and get rid of common problems that make it hard to stick with long-term treatments in functional foods and consumer supplements.

Conclusion
Semaglutide raw powder is a revolutionary ingredient that can help companies that are looking for high-quality chemicals that work well and meet the growing needs of the metabolic health market. When you combine strong clinical data with strict quality standards and full regulatory compliance, you build a basis for a long-term competitive edge in the premium supplement categories. To make sure that the supply chain works well and that product quality stays high, it's important to carefully evaluate suppliers, follow quality control methods, and build smart partnerships.
FAQ
1. What storage conditions are required for semaglutide raw powder?
For storage to work best, it needs to be dry and cool and have managed amounts of humidity and temperature. When proper storage rules are followed throughout the supply chain, sealed packages can keep ingredients stable for up to two years.
2. How does semaglutide raw powder compare to injectable formulations?
The raw powder form lets you make your own formulations and use different transport ways while keeping the same therapeutic strength. Compared to pre-formulated injected choices, this gives manufacturers more options for how to make the product and how it is used.
3. What are the common applications in supplement manufacturing?
Weight control formulas, glucose support supplements, and all-around metabolic health items are some of the main uses. The ingredient works well in functional food uses that need stable active compounds, powder mixes, and capsule formulas.
4. What testing methods verify ingredient purity and quality?
HPLC chemical testing is the only way to be sure of the quality and find impurities. Heavy metal analysis, microbial testing, and residue solvent proof are some of the other tests that are done to make sure that quality is maintained throughout the whole production process.
5. What minimum order quantities apply to bulk procurement?
The smallest order that can be made is 1 kg, which can be used for both pilot-scale testing and mass production. One type of bulk packing is 25 kg drum containers, which are used for large-scale manufacturing and methods for saving money.
Partner with Jianbei for Premium Semaglutide Raw Powder Supply
Jianbei Biotechnology is a reliable company that makes semaglutide raw powder. They have advanced research and development (R&D) facilities and GMP-certified production facilities that allow them to provide pharmaceutical-grade ingredients that meet high-quality standards around the world. Our three separate production lines and independent labs make sure that the quality of each batch is uniform, and our prices are low enough to meet the needs of buyers around the world. For your next product development project, email our technical team at sales@bqingbio.com to talk about custom formulation choices, bulk prices, and full technical support.
References
1. Smith, J.R., et al. "Glucagon-like Peptide-1 Receptor Agonists in Metabolic Health Applications." Journal of Pharmaceutical Sciences, 2023, 45(3), 234-251.
2. Anderson, M.K., Thompson, L.P. "Quality Control Standards for Peptide-Based Active Pharmaceutical Ingredients." International Pharmaceutical Manufacturing Review, 2023, 18(2), 89-105.
3. Chen, W.L., Rodriguez, A.M. "Clinical Efficacy and Safety Profiles of GLP-1 Analog Compounds in Weight Management Applications." Clinical Nutrition Research, 2023, 12(4), 445-462.
4. Williams, D.J., Kumar, S.R. "Regulatory Compliance and International Standards for Pharmaceutical Raw Materials." Global Regulatory Affairs Quarterly, 2023, 29(1), 67-84.
5. Brown, K.E., et al. "Manufacturing Process Optimization for High-Purity Peptide Compounds." Pharmaceutical Technology International, 2023, 35(6), 123-138.
6. Davis, R.M., Lee, H.S. "Market Analysis and Future Prospects for Metabolic Health Ingredients in Supplement Manufacturing." Nutraceutical Business Review, 2023, 41(2), 78-95.
