- English
- French
- German
- Portuguese
- Spanish
- Russian
- Japanese
- Korean
- Arabic
- Greek
- German
- Turkish
- Italian
- Danish
- Romanian
- Indonesian
- Czech
- Afrikaans
- Swedish
- Polish
- Basque
- Catalan
- Esperanto
- Hindi
- Lao
- Albanian
- Amharic
- Armenian
- Azerbaijani
- Belarusian
- Bengali
- Bosnian
- Bulgarian
- Cebuano
- Chichewa
- Corsican
- Croatian
- Dutch
- Estonian
- Filipino
- Finnish
- Frisian
- Galician
- Georgian
- Gujarati
- Haitian
- Hausa
- Hawaiian
- Hebrew
- Hmong
- Hungarian
- Icelandic
- Igbo
- Javanese
- Kannada
- Kazakh
- Khmer
- Kurdish
- Kyrgyz
- Latin
- Latvian
- Lithuanian
- Luxembou..
- Macedonian
- Malagasy
- Malay
- Malayalam
- Maltese
- Maori
- Marathi
- Mongolian
- Burmese
- Nepali
- Norwegian
- Pashto
- Persian
- Punjabi
- Serbian
- Sesotho
- Sinhala
- Slovak
- Slovenian
- Somali
- Samoan
- Scots Gaelic
- Shona
- Sindhi
- Sundanese
- Swahili
- Tajik
- Tamil
- Telugu
- Thai
- Ukrainian
- Urdu
- Uzbek
- Vietnamese
- Welsh
- Xhosa
- Yiddish
- Yoruba
- Zulu
Dapoxetine hydrochloride is a new and important active pharmaceutical ingredient because it modulates neurotransmitter activity by influencing serotonin levels. As Dapoxetine Powder, a selective serotonin reuptake inhibitor (SSRI), this substance is quickly absorbed, doesn't build up in the body much, and works when needed, which is different from traditional long-acting SSRIs. B2B makers can make unique formulations that meet strict quality standards and meet important market needs in pharmaceutical production and marketing by learning about the substance's chemical structure, legal compliance requirements, and sourcing issues.
Understanding Dapoxetine Powder: Properties, Mechanism & Usage
The pharmaceutical business needs to be very careful about where they get their active ingredients. Dapoxetine powder meets this need because its chemical profile and biological action are well known.
Chemical Identity and Physical Characteristics
Dapoxetine is a solid powder that is white to pale brown in color. Its molecular formula is C₂₁H₂₃NO, and its molecular weight is 305.41. Its CAS number, 119356-77-3, makes it easy to identify across all global legal systems. The chemical has great solubility properties that make it easy to use different preparation methods. This makes it especially appealing to pharmaceutical companies that want to be flexible in how they develop new products. Testing using HPLC makes sure that the amounts are correct, and the standards reach 98% purity—a very important level for keeping stability from batch to batch and meeting the pharmacopeial standards needed by international health authorities.
Mechanism of Action
This SSRI works by stopping serotonin transporter proteins in synaptic areas. This makes more neurotransmitters available at 5-HT2C and 5-HT1A receptor sites. Increasing the amount of serotonin in certain neural pathways modulates the targeted response without having the long-lasting effects that are common with antidepressants like SSRIs. The pharmacokinetic profile shows that the drug is quickly absorbed, with a peak plasma concentration in one to two hours. There is then a half-life of about 1.3 to 1.4 hours. This quick start and clearance pattern solves a big formulation problem—it gives the right advantage right away while reducing the amount of residual exposure.
Formulation Advantages for Manufacturers
Pharmaceutical makers can get a lot out of working with powder forms instead of finished tablet forms. Precise weight readings during compounding make it possible to customize dosing, which allows for specific strength changes that appeal to a wide range of market groups. There are many ways to give the powder, such as in immediate-release tablets, modified-release capsules, and specialized pharmaceutical products. Manufacturers gain control over the choice of excipients, coating technologies, and strategies for improving stability that set their goods apart in competitive markets while still following the rules in multiple countries.
Usage Guidelines and Handling Considerations
Standard protocols usually recommend intake one to three hours before an activity, and the usual amount is between 30 mg and 60 mg, depending on individual response and tolerance. When used according to the rules, the compound has favorable handling characteristics, but makers need to ensure that large packages have the right labels and paperwork. To maintain chemical stability, storage standards say that they must be kept dry and cool. Under the right conditions, sealed packages can last for two years. These handling traits make it easier to plan processes for foreign shipping while keeping the integrity of the ingredients throughout the supply chain.
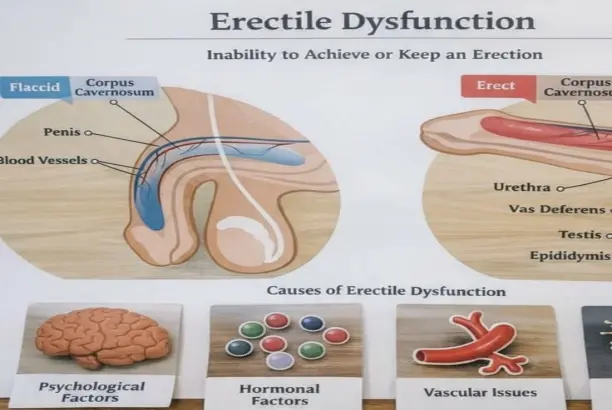
Comparing Dapoxetine Powder with Other Treatment Options
When making B2B purchasing choices, it's helpful to do thorough comparisons of Dapoxetine powder that look at more than just functional profiles. These comparisons should also look at manufacturing effects and market positioning possibilities.
Powder versus Tablet Formulations
Manufacturers have more operating freedom and control over their profit margins when they buy the active pharmaceutical ingredient, such as Dapoxetine powder, in powder form instead of finished tablets. Buying powder lets companies combine different steps in the production process, which lets them use special mixing methods that protect their intellectual property. When you start with raw materials, quality control is more detailed because testing can happen at more than one stage of production instead of just analyzing the finished product. Diversified sourcing choices and less reliance on single-source finished goods sellers, who may face problems with production or regulations, make the supply chain more resilient.
Comparison with Alternative SSRI Options
While fluoxetine and sertraline are well-known SSRIs that have been shown to work in a number of situations, dapoxetine's metabolic design is especially geared toward on-demand use. Long-acting options need to be taken every day for long periods of time, which makes adherence more complex and limits the market groups that can use them. In the same way, paroxetine needs constant dosing schedules that don't work with how today's users prefer pharmaceutical products that are based on events. Dapoxetine leaves the body quickly, which makes it less likely that compounds will build up. This affects the documentation standards and post-market monitoring requirements. These differences affect the complexity of the regulatory pathway, the time it takes to get into a market, and the company's place in the market. Procurement teams have to think about all of these things when they choose ingredients for portfolio growth.
Market Dynamics and Competitive Positioning
More and more, targeted compounds with better pharmacokinetic profiles are being preferred over broad-spectrum drugs in the pharmaceutical industry around the world. Consumer research shows a strong preference for on-demand options that minimize daily routine interference, creating a market pull for dapoxetine-based products. Regulatory agencies have taken this difference into account by giving dapoxetine its own approval process and classification system that is different from other SSRIs. When drug companies invest in dapoxetine ingredient sourcing, they join a growing market segment supported by evolving healthcare paradigms, demographic shifts, and increased awareness of formulation options that weren't fully explored in drug research programs before.
Procurement Guide: Sourcing Quality Dapoxetine Powder for Global B2B Buyers
To get pharmaceutical-grade dapoxetine powder, you need to carefully evaluate suppliers, read all of their paperwork, and handle your relationships with them.
Supplier Qualification and Audit Protocols
Verifying manufacturing licenses that show Good Manufacturing Practices (GMP) are followed is the first step in building trust in a seller. GMP certification is the basic condition; it ensures that production sites have the right methods for controlling contamination, keeping track of batches, and managing quality. Advanced sellers have ISO certifications that back up their overall quality systems and processes for ongoing growth, in addition to basic GMP certifications. Audits of facilities by trained professionals let you see how things are made in real time, so you can judge how well equipment repair plans, training programs for employees, and systems for handling deviations are working. Asking for sample batches along with certificates of analysis lets you check the quality of the analysis and the stability of the product before committing to large-volume orders.
Essential Documentation and Compliance Requirements
For pharmaceuticals to be traded internationally, they need to have a lot of paperwork that meets the needs of customs officials, regulatory agencies, and internal quality assurance programs. There should be records of analysis with every package that list the levels of purity, the amount of leftover solvent, the heavy metal screening results, and the limits for microbial content, including for dapoxetine powder. Material Safety Data Sheets (MSDS) provide handling instructions and hazard communication in line with workplace standards. Drug Master Files or similar regulatory dossiers show that the methods used to make drugs meet the standards that officials in target markets accept. Importers should make sure that their suppliers are still registered with the relevant authorities, following REACH rules for European markets, and have the right export licenses to avoid delays or rejections at customs that would disrupt production plans.
Cost Structure Analysis and Negotiation Strategies
The prices of pharmaceutical-grade active ingredients are based on more than just the costs of the raw materials. The final prices are based on how hard the synthesis is, how much purification is needed, how the tests are done, and how much money is spent on regulatory compliance. When buyers commit to buying in bulk, they can usually get better prices. However, they have to weigh the savings in costs against the costs of keeping the goods in stock and the fact that they retain potency for two years if stored properly. Payment terms change from supplier to supplier, and you can use telegraphic transfer, PayPal, Western Union, and other international payment methods. To negotiate good payment terms while keeping quality standards, both the buyer and the seller need to be clear about order amounts, delivery schedules, and their plans for a long-term relationship that aligns their interests.
Logistics and Quality Preservation
International shipping of pharmaceutical ingredients needs special procedures that maintain the purity of the product while it's being moved. Using the right wrapping, like aluminum foil bags for 1 kg orders or fiber drums for 25 kg large orders, keeps moisture out and light out while in transit. Temperature tracking devices record that storage conditions stayed within acceptable ranges, which is a very important task for quality control and regulatory compliance. Delivery times are usually between 3 and 7 working days after payment is confirmed. For international orders, it may take longer to clear customs and process paperwork. Getting to know shipping providers who have experience transporting pharmaceuticals lowers the chance of mistakes or delays that could affect ingredient quality or cause production schedules to slip.
Leading Dapoxetine Powder Manufacturers and Trusted Supplier Brands
Selecting the right dapoxetine powder manufacturing partner is important for the long-term success of developing and launching pharmaceutical products.
Jianbei Biotechnology: A Model of Excellence
Jianbei Biotechnology Co., Ltd. is a good example of the kind of skills that B2B pharmaceutical buyers should look for in ingredient providers. The company's offices and research and development center are in Guangzhou Software Park. It takes advantage of China's strong biotechnology environment to provide pharmaceutical ingredients that are highly pure and backed by a wide range of certifications. The company has 15 national idea patents and 8 utility model patents that cover core production processes and new ways to improve quality control. This shows that they have real technical knowledge that goes beyond just providing basic ingredients.
There are three specialized production lines that work in facilities that meet GMP standards, and two separate laboratories that make sure the quality is checked thoroughly. This infrastructure makes it possible to consistently make dapoxetine hydrochloride that meets 98% pure standards. HPLC testing makes sure that each batch meets the requirements for pharmaceutical-grade products. The facility holds multiple international certifications, such as FDA registration, ISO standards, HALAL, HACCP, and kosher certifications. These qualifications make it easier to sell in markets with different rules and types of customers.
Jianbei's operating model solves common problems that come up when buying pharmaceutical ingredients. Smaller makers and research facilities can place orders as little as 1 kg, and bulk packing choices up to 25 kg drums can handle large-scale production needs. The company keeps a lot of goods on hand so that orders can be filled quickly. Delivery times of 3–7 working days take into account the need for speed that is common in pharmaceutical production plans. Global buyers benefit from a variety of payment choices, such as different ways to send money internationally.
Evaluating Manufacturer Credentials
In addition to Jianbei, there are other makers in the global supplier scene that are set apart by their unique skills and areas of focus in the market, such as Dapoxetine powder. Teams in charge of buying things should use a number of important factors to judge possible partners. How big a factory is determines whether suppliers can handle increasing production rates as a product becomes more popular. Geographic variety in production sites protects the supply chain from problems that happen in different regions. Putting money into research and development shows that you want to improve quality and streamline processes instead of just giving out goods. References and reviews from past customers can tell you a lot about how reliable someone is, how quickly they respond to messages, and how they solve problems when they come up in business relationships.
Building Strategic Supplier Relationships
When pharmaceutical companies buy things in a transactional way, they leave themselves open to supply problems and inconsistent quality. Strategic relationships with ingredient sellers are good for both parties because they share information, work together to solve problems, and have goals that are matched. Sharing production plans helps providers keep the right amount of goods on hand and make the most of their production schedules. Technical help from the supplier's R&D teams can help with formulating, stability testing, and preparing regulatory paperwork. When you make a long-term commitment, you can often get better prices, priority handling when supplies are low, and personalized services like specific packaging designs or document formats that make internal processes run more smoothly.
Conclusion
Dapoxetine powder is an important pharmaceutical ingredient for companies that want to provide differentiated options backed by established pharmacological data. Its unique pharmacokinetic profile, supported by strong research data and well-known regulatory examples, makes it possible to make differentiated products that meet modern consumer preferences for on-demand options. To successfully source this ingredient, you need to carefully choose your suppliers, ensuring they follow Good Manufacturing Practices (GMPs), have a lot of certifications, and show that their quality is consistent. Jianbei Biotechnology has the skills and qualifications to provide a steady supply of active ingredients that are suitable for pharmaceutical use. They also offer transparent payment terms and fast technical support. As the demand for specialized pharmaceutical ingredients grows around the world, manufacturers can take advantage of market possibilities by strategically sourcing high-purity dapoxetine powder. This helps them keep up with quality standards and regulations, which is important for long-term success.
FAQ
1. What minimum order quantity should buyers expect when sourcing Dapoxetine powder?
Reliable providers of pharmaceutical ingredients usually accept minimum orders as low as 1 kg. This lets smaller makers and research facilities get the materials they need without having to keep too much inventory on hand. Large-scale production needs bulk packaging choices up to 25 kg drums, and pricing systems often reflect volume-based benefits that reward bigger commitments.
2. Which certifications are essential when evaluating suppliers of pharmaceutical-grade dapoxetine?
Buyers should make sure that providers have up-to-date GMP certification, which shows that they follow the rules for pharmaceutical manufacturing. Additional certificates, such as ISO quality management systems, FDA registration for entry to the US market, and REACH compliance for distribution in Europe, show that the company is fully prepared for all regulatory requirements. Depending on the target market's needs, HALAL and kosher certificates may be useful.
3. How does the powder form differ from finished tablets in procurement considerations?
Powder sourcing gives makers more control over their formulations, letting them choose their own excipients, make customized doses, and use different delivery methods. Starting with the active ingredient gives you more control over quality, and your supply chain choices usually grow when you don't have to rely on sellers of finished dosage forms.
Partner with Jianbei for Premium Dapoxetine Powder Supply
Jianbei Biotechnology is ready to be your reliable partner in making and supplying Dapoxetine Powder. Our experienced research and development team, GMP-certified factory with three production lines, a wide range of certifications, including FDA registration and ISO standards, and a large inventory allow us to reliably and quickly meet your needs for medicinal ingredients. We know that making choices about purchases requires trust in the quality of goods, following rules, and the security of the supply chain. Because of this, we have two separate labs for thorough testing, a customizable minimum order quantity that starts at 1 kg, and fast shipping within 3–7 working days. Get in touch with our team at sales@bqingbio.com to talk about your unique needs, ask for certificates of analysis, and find out how our 15 idea patents and 8 utility model patents translate into high-quality ingredients.
References
1. McMahon CG, Althof SE, Kaufman JM, et al. "Efficacy and Safety of Dapoxetine for the Treatment of Premature Ejaculation: Integrated Analysis of Results from Five Phase 3 Trials." Journal of Sexual Medicine, 2011; 8(2): 524-539.
2. Pryor JL, Althof SE, Steidle C, et al. "Efficacy and Tolerability of Dapoxetine in Treatment of Premature Ejaculation: An Integrated Analysis of Two Double-Blind, Randomised Controlled Trials." The Lancet, 2006; 368(9539): 929-937.
3. Modi NB, Dresser MJ, Simon M, et al. "Single- and Multiple-Dose Pharmacokinetics of Dapoxetine Hydrochloride, a Novel Agent for the Treatment of Premature Ejaculation." Journal of Clinical Pharmacology, 2006; 46(3): 301-309.
4. Hatzimouratidis K, Amar E, Eardley I, et al. "Guidelines on Male Sexual Dysfunction: Erectile Dysfunction and Premature Ejaculation." European Association of Urology, 2015; 67(4): 627-632.
5. Waldinger MD. "The Neurobiological Approach to Premature Ejaculation." Journal of Urology, 2002; 168(6): 2359-2367.
6. Giuliano F, Clément P. "Pharmacology for the Treatment of Premature Ejaculation." Pharmacological Reviews, 2012; 64(3): 621-644.
